Targeting glioma stem cells via the Hedgehog signaling pathway
Abstract
Cancer is one of the leading causes of death worldwide. Gliomas are among the most devastating tumor types, and current clinical therapies are unsatisfactory. Recent reports revealed the importance of glioma‑propagating cells in the malignancy of gliomas. These cells, also referred to as glioma stem cells (GSCs), share similarities with neural stem cells (NSCs). The Hedgehog (Hh) signaling pathway controls tissue polarity, patterning maintenance, and maintenance of NSCs during embryonic development. Aberrant activation of the Hh pathway resulting from mutation and deregulation has recently been recognized to cause tumorigenesis in a wide variety of tissues, including gliomas and GSCs. In this review, we explore the role of the Hh signaling pathway in GSCs and its potential as a therapeutic strategy.
Keywords
Introduction
Cancer is estimated to be the leading cause of death worldwide by the World Health Organization (WHO).[1] Gliomas are one of the most lethal adult brain tumors. Although substantial progress has been made, there is still a lack of effective therapy. It is reported that patients with low-grade gliomas can survive for years, while patients with glioblastoma (WHO grade IV) survive for only 1-2 years after diagnosis.[2]
Tumors are composed of a heterogeneous group of cells. Some tumor cell fractions have the ability to initiate tumors in xenograft models, whereas other fractions do not.[3] These cells, capable of sphere-like growth in vitro and tumor formation in vivo, are defined as cancer stem cells (CSCs) and share similarities with normal neural stem cells (NSCs). It has been hypothesized that these cells are involved in radio- and chemo-resistance, as well as tumor recurrence.
The Hedgehog (Hh) signaling pathway plays a pivotal role in embryonic development, including the formation and maintenance of glioma stem cells (GSCs).[4] Since GSCs are important biological factors responsible for cancer invasion, metastasis, drug resistance, and relapse, Hh signaling is believed to be an important target for cancer therapy. Recently, both natural and synthesized small-molecule inhibitors of Hh signaling have been investigated as potential cancer treatments. However, targeting only one molecule may be insufficient. Therefore, strategies using a combination of natural products and chemotherapeutics with different targets may improve the overall survival of patients with gliomas.
Cancer stem cells in glioma
Cancer stem cells and the niche
The concept of CSCs has been in use for over 50 years. In the 1990s, Lapidot et al.[5] identified leukemia stem cells capable of generating human acute myeloid leukemia after transplantation. Within gliomas, stem- like cells with the ability to self-renew, differentiate into multiple lineages, and initiate tumors are known as GSCs. Subsequently, GSCs capable of self-renewal and producing glioma-initiating cells were identified using a limiting dilution analysis.[6] The presence of GSCs and the increasing evidence of radio-resistance and chemo- resistance indicated that GSCs may contribute to tumor maintenance and recurrence, and that targeting GSCs may be a promising therapeutic intervention.[7,8] Stem cells reside in specialized niches, which could regulate their proliferation and differentiation.[9] More than one study has demonstrated the presence of GSCs near blood vessels, consistent with the perivascular niche for NSCs.[10,11] Alternatively, Li et al.[12] demonstrated that GSCs could also be found in regions of necrosis, which are hypoxic, suggesting that there may be more than one niche. Hypoxia promotes the self-renewal capability of both the stem cell and nonstem cell populations, and also promotes the conversion of nonstem cells into stem cells by up-regulating the expression of important stem cell factors,[13] indicating that this niche may be important for GSCs. Therapies that target cytokines, such as hypoxia-inducible factor- 1α (HIF-1α) and HIF-2α, or cells in the niche, such as glia cells and endothelial cells, may, therefore, show promise.
Glioma stem cells and resistance to therapy
Surgical resection, radiation, and chemotherapy are still the mainstay treatments for gliomas and are associated with a clear improvement in overall survival in patients with high-grade gliomas. However, recurrence of the tumor is common following conventional therapy. Antiangiogenic therapy against vascular endothelial growth factor is another frequently used therapeutic treatment, but drug resistance is common.[14] The mechanisms of resistance are not understood in complete detail and may be multi-factorial. The proposal that GSCs are a prerequisite for tumor formation suggests that chemo- and radio-resistant GSCs are the main cause of recurrence.[15,16] Liu et al.[17] found that CD133+ cells were resistant to chemotherapeutic agents, whereas CD133− cells sorted from the same primary glioma cultures were not. Furthermore, Bao et al.[7] determined that ionizing radiation treatment enriched the CD133+ population in human gliomas. Although CD133 does not identify all GSCs, these data suggest that GSCs play a key role in resistance to traditional therapies. The mechanism for resistance is complicated. Bao et al.[7] demonstrated that CD133+ cells contribute to glioma radio-resistance via preferential activation of DNA damage checkpoints, and that their resistance could be partially reversed by inhibitors of Chk1 and Chk2. Other researchers demonstrated that the bone morphogenetic proteins and cannabinoids inhibit the tumorigenic potential of GSCs and promote their differentiation.[18] Radiation treatment may expand the GSC population, enhance the aggressiveness of tumors, and induce expression of GSC marker proteins, such as CD133 and nestin, as well as proteins involved in self-renewal, such as Notch2 and Sox2.[19] The GSC niche may enhance the radio-resistance of GSCs as well.[20] Besides their relative radio-resistance, GSCs can express high levels of multiple genes associated with drug resistance[21] and show significant resistance to chemotherapeutic agents.[17] Temozolomide (TMZ) treatment, which could eliminate O6-methylguanine- DNA methyltransferase-negative cells, increased the stem population.[22] Increased expression of drug transporters, such as ATP-binding cassette (ABC) transporters, could lead to chemotherapeutic agents being pumped out of tumor cells and may be another mechanism of chemo-resistance. Recent studies have found that the expression of ABC transporters is increased in stem cells.[23] Therefore, focusing on the connection between GSCs and their niche may help to elucidate the mechanisms behind the treatment-resistant phenotype of GSCs, which may lead to solutions that reduce the resistance of gliomas to therapy and improve clinical outcome. This information may also be useful in the pursuit of effective therapeutic strategies for the treatment of radiation-associated injuries.
Cell sorting and culture
Several methods may be used in order to obtain purified GSCs for study. In some studies, fluorescence-activated cell sorting and magnetic activated cell sorting have been used to separate GSCs from other cell types.[12,24] Glioma cell lines and clinical samples have also been used to isolate and culture GSCs.[25,26] However, there is currently no standard definition of what constitutes a GSC. Specific markers for identifying GSCs are, therefore, required. The current definition of GSCs is based on their capacity for self-renewal, long-term proliferation, and tumor formation in vivo. The ideal marker, therefore, is a molecule that is specifically expressed on GSCs and functionally associated with GSC maintenance.
CD133 has been widely used as a marker for GSC sorting. However, NSCs also express CD133, which limits its utility and reliability as a target. Some studies have also suggested that CD133− cells have the capability to act as GSCs.[13] Moreover, CD133− glioma cells can give rise to CD133+ GSCs.[13] Other surface molecules such as CD15 (SSEA-1),[27] A2B5,[28,29] and L1CAM[30] have been used as markers but are not widely accepted. Interestingly, both CD133+ A2B5− and CD133− A2B5+ cells have been shown to exhibit characteristics of GSCs.[31] Therefore, these markers may only label specific sub-populations of GSCs. In recent times, one group exploited the intrinsic auto-fluorescence properties and distinctive morphology of a subpopulation of cells (FL1+) in order to isolate them from human gliomas. FL1+ cells are capable of self-renewal in vitro, tumorigenesis in vivo, and preferentially express stem cell genes, but expression of FL1 did not correlate with the expression of other proposed GSC markers.[32] This finding deserves special attention as it may provide a new way to identify GSCs.
Hedgehog in glioma
Hedgehog signaling pathway
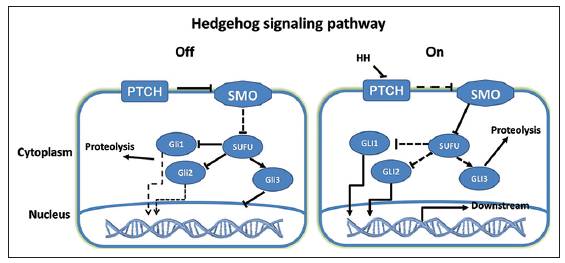
Hedgehog is a highly conserved signaling pathway and a key regulator of embryonic development, including the processes of cell differentiation, proliferation, and tissue patterning.[33,34] In adults, Hh plays an important role in the maintenance of stem cells, tissue repair, and regeneration. The Hh family consists of three secreted proteins, sonic Hedgehog (Shh), Indian Hedgehog (Ihh) and desert Hedgehog. Two molecules that are important for Hh signaling are patched (Ptch) and smoothened (Smo). In the absence of Hh, Ptch inhibits the activity of Smo, a receptor-like protein with seven transmembrane domains. In the presence of activated Smo, a complex consisting of the glioma-associated oncogene family zinc finger (Gli) and the suppressor of fused (Sufu), an important negative regulator of Hh signaling, enters the nucleus, leading to nuclear translocation and activation of the Gli1 and Gli2 transcription factors, as well as degradation of Gli3. Activated Gli subsequently promotes the transcription of Hh target genes [Figure 1]. Three types of Gli transcription factors, Gli1, Gli2, and Gli3, have been identified in mammals. Gli1 and Gli2 are activators of Hh target genes, while Gli3 mainly appears to be a repressor. The function of Hh signaling is very complicated and critical. Thus, it is important to elucidate the function and molecular regulation of Hh signaling, especially in GSCs.
Figure 1. The off and on states of the Hedgehog (Hh) signaling pathway. Patched (Ptch) inhibits the activity of smoothened, which prevents the suppressor of fused-Gli1/2 complex from entering the nucleus and promotes Gli3 nuclear accumulation, leading to low expression of Hh target genes. Hh binding to Ptch activates the Hh signaling pathway by promoting Gli1/2 expression
Functional studies of Hedgehog in glioma stem cells
Aberrant activation of Hh signaling has been shown to be associated with the formation of gliomas. Several studies have investigated the role of Hh-Gli signaling in GSCs and found that it regulates self-renewal and tumorigenic potential in GSCs.[22,35-37] Importantly, inhibition of Hh-Gli signaling enhances the ability of TMZ to inhibit GSC proliferation and induce cell death.[38] Several studies have demonstrated that inhibition of Hh signaling blocks tumor growth and influences both proliferation and malignancy.[39] The Shh pathway plays an important role in the migratory ability of cells derived from CD133− glioblastoma cells.[35] Furthermore, the Hh inhibitor cyclopamine has been shown to improve the effect of radiation on GSCs. All of the studies mentioned above suggest that Hh signaling is one of the critical pathways for the maintenance, proliferation, migration, and tumorigenic potential of GSCs. Thus, targeting this pathway with pharmacologic inhibitors may inhibit GSC growth and improve the efficacy of conventional therapies.
The regulatory role of microRNA
MicroRNAs (miRNAs) are a class of small noncoding cellular RNAs that bind to cis-regulatory elements located primarily in the 3’ untranslated regions of target mRNAs, resulting in their translational inhibition or degradation. The function of some miRNAs has been determined to be important for neural development.[40] Other studies have indicated that miRNAs play a potentially important role in glioma biology. The relationship between the Hh pathway and miRNA is currently being investigated.
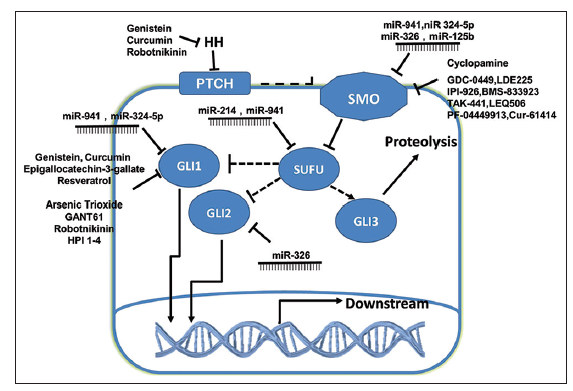
It has been shown that stable miR-302-367 cluster expression is sufficient to suppress the stem cell-like signature, self-renewal, and infiltration of cells by inhibition of the CXCR4 pathway. Furthermore, inhibition of CXCR4 leads to disruption of the Shh-Gli-Nanog network, which is involved in self-renewal and expression of the embryonic stem cell- like signature.[41,42] Wu et al.[43] suggested that miR-5 can specifically suppress Hh signaling by directly targeting Smo in Drosophila. In addition, miR-125b and miR-326 have been identified as suppressors of Smo, and miR- 324-5p targets the downstream transcription factor Gli1. Down-regulation of these miRNAs allows high levels of Hh-dependent gene expression leading to tumor cell proliferation.[44] Furthermore, functional analyses have shown that miR-326 may be regulated by Shh activation and act as a negative modulator of Shh signaling by directly targeting Smo and Gli2.[45] Other studies have demonstrated that miR-214 can inhibit Sufu, allowing maximal activation of Gli-mediated transcription,[46] and miR-941 targets key components of the Hh-signaling pathway, including Smo, Sufu, and Gli1.[47] Moreover, miR-212 was found to be involved in tumorigenesis, and the oncogenic activity of miR-212 was partly due to suppression of Ptch1.[48] Although many miRNAs have been shown to regulate the Hh pathway as upstream factors, the Hh pathway in turn has been shown to regulate the mir-29b-1/mir-29a promoter [Table 1 and Figure 2].[49]
MicroRNAs and their targets in Hh signaling pathway
| MicroRNA | Target |
|---|---|
| miR-302~367 | CXCR4 Shh-Gli-Nanog |
| miR-125b | Smo |
| miR-326 | Smo, Gli2 |
| miR-324-5p | Smo, Gli1 |
| miR-214 | Sufu |
| miR-941 | Smo, Sufu, Gli1 |
| miR-212 | Ptch1 |
The studies mentioned above show that miRNAs affect the expression of numerous genes involved in glioma biology. Identifying the roles that different miRNAs play may help to understand the mechanisms leading to glioma-propagation and provide new therapeutic strategies. In particular, miRNAs that affect the Hh pathway should be investigated in more detail.
Cross‑talk with other pathways
Cross-talk between the Hh signaling pathway and other embryonic signaling pathways, such as the Notch and Wnt pathways, has been reported not only in glioma cell lines, but in other cancers as well. Cross- talk between signaling pathways has the potential profoundly to add to the complexity of cellular responses to external stimuli. Wnt signaling directs the development of a variety of organ systems during embryogenesis. In adults, Wnt signaling has a key role in the regulation of tissue self-renewal. Over the past several years, various discoveries have suggested that there are fundamental similarities between the Wnt and Hh signaling pathways.[50] Both pathways are activated by a membrane protein (Frizzled or Smo) and prevent phosphorylation-dependent proteolysis of key effector (β-catenin or cubitus interruptus), which converts a DNA-binding protein from a repressor into an activator of transcription. In addition, silencing of both pathways in the absence of ligand requires Slimb-β-TRCP-FWD-1, which is a component of the SCF ubiquitin ligase complex, and the protein kinases glycogen synthase kinase 3 (GSK3) and casein kinase 1 (CK1). It has been observed that molecules involved in Wnt signaling, such as GSK-3 β, regulate the Hh signaling pathway.[51] In turn, activation of Gli may stimulate the transcription of Wnt ligands. It has been found that GSK-3 β phosphorylates and stabilizes Sufu, leading to inhibition of Hh activation.[52] Moreover, the Hh pathway was found to inhibit Wnt signaling, as a result, of Gli1 induction through up-regulation of secreted frizzled-related protein 1.[53]
Notch signaling is another conserved developmental signaling pathway that is important for embryogenesis, cellular homeostasis and stem cell renewal.[54] Notch receptor activation induces the expression of hairy and enhancer of split 3 (Hes3) and Shh through rapid activation of cytoplasmic signals, including the serine/threonine kinase Akt, the transcription factor STAT3, and the mammalian target of rapamycin (mTOR), leading to the promotion of NSC survival.[55] Simultaneously, Shh induces the expression of another specific target gene, Hes1, and Smo function has been found to be necessary for Shh-induced up- regulation of Hes1.[56] Moreover, inhibition of Shh and Notch may enhance the sensitivity of CD133+ GSCs to TMZ;[38] and the Shh, Notch, and Wnt pathways combined may regulate self-renewal and differentiation of breast CSCs and progenitor cells.[57]
Aberrant activation of epidermal growth factor receptor (EGFR) signaling has been implicated in a number of human malignancies, which has made EGFR a prime molecular target in chemotherapy.[58] Several studies suggest that the combination of specific EGFR and Hh inhibitors may provide a therapeutic benefit. For instance, the combination of the selective EGFR inhibitor gefitinib and the Smo antagonist cyclopamine, with or without the chemotherapeutic drug docetaxel, inhibits cell growth and induces apoptosis.[59] EGFR synergizes with Gli1 and Gli2 to selectively activate transcription of Gli target genes via stimulation of RAS/RAF/MEK/ERK signaling.[60] Moreover, EGF has already been shown to have the capability to stimulate the proliferative activity of Shh on NSCs and to enhance the invasive properties of epidermal cells expressing Shh.[61,62] Further investigation is required, however, in order to understand these interactions in more detail.
Hedgehog signaling may also be involved in cross- talk with other pathways, such as transforming growth factor-β (TGF-β) and AKT signaling. TGF-β has been shown to promote Gli2-mediated expression of parathyroid hormone-related protein.[63] Other studies have indicated that co-activation of the Hh and AKT pathways promote tumorigenesis.[64] The results of these studies, in addition to those mentioned above, indicate the existence of cross-talk between Hh signaling and several other pathways. It is, therefore, possible that combination therapies targeting both Hh and other pathways may provide additional benefits to patients in comparison with individual treatments.
Hedgehog as a therapeutic target in glioma
In recent times, several inhibitors of Hh signaling have been synthesized or discovered for use in studies of cancer treatment in vitro and in vivo. By targeting important molecules in the Hh signaling pathway, Hh inhibitors down-regulate the activity of Hh signaling in cancer cells, resulting in the inhibition of cancer cell growth and tumor progression. GDC-0449 (vismodegib) is a small-molecule inhibitor specifically designed to target Smo. In preclinical experiments, GDC-0449 has been shown to inhibit the activation of the Hh pathway, leading to the inhibition of tumor growth initiated by mutations of Ptch or by increased levels of Hh ligands.[65] A search of the clinicaltrials.gov database identified 38 clinical trials of GDC-0449 focused on the treatment of different malignancies. In one trial, for example, GDC-0449 was tested in combination with Avastin (bevacizumab) and traditional chemotherapy in metastatic colorectal cancer. Treatment with GDC-0449 resulted in a reduction of symptoms, and the data suggests that GDC-0449 may be safely used in combination with conventional agents. Unfortunately, mutations in Smo and its downstream targets are common, and may lead to GDC-0449 resistance. However, it has been shown that resistant medulloblastomas are sensitive to PI3K inhibition, which may indicate that combined therapy is necessary.[66]
LDE225, another Hh inhibitor specifically targeting Smo, has also been shown to reduce Hh-dependent proliferation. The main side effects include nausea, vomiting, anorexia, fatigue, muscle cramps, and dysgeusia. During the course of LDE225 treatments, resistance to the drug was observed. Possible mechanisms for this resistance include Gli2 amplification and Smo mutations, leading to reactivation of Hh signaling. Similar to the GDC-0449 study, a combination treatment of LDE225 with PI3K inhibitor delayed the development of resistance.[67] Thus, combined therapy targeting multiple pathways needs more investigation.
IPI-926 (saridegib) is a unique, selective, and potent molecule that inhibits Smo. IPI-926 is orally bioavailable and has demonstrated biological activity in multiple preclinical animal models of cancer. IPI-926 appears to down-regulate Hh signaling, leading to inhibition of the potential for self-renewal. Drug resistance was observed after extended treatment periods, but was primarily caused by increased expression and activity of P-glycoprotein drug transporters rather than the emergence of genetic mutations that prevent drug-target interactions.[68] BMS-833923 is another Hh inhibitor that acts by binding to Smo. Clinical trials have been conducted to evaluate the effects, safety, tolerability, and pharmacokinetics of BMS-833923 alone or in combination with other drugs. Resistance to this drug and the mechanisms behind it still need to be studied, however. In addition, other synthetic Smo antagonists including TAK-441, LEQ506, PF-04449913, and Cur-61414 have already been tested in clinical trials in order to determine dosage levels and evaluate safety [Table 2 and Figure 2].
Synthetic inhibitors of Hedgehog signaling pathway
| Synthetic inhibitors | Target |
|---|---|
| GDC-0449 (Erivedge, vismodegib) | Smo |
| LDE225 | Smo |
| IPI-926 (Saridegib) | Smo |
| BMS-833923 | Smo |
| TAK-441 | Smo |
| LEQ506 | Smo |
| PF-04449913 | Smo |
| Cur-61414 | Smo |
| Arsenic trioxide | Gli |
| GANT61 | Gli |
| Robotnikinin | Shh |
| HPI 1-4 | Gli |
Figure 2. Inhibition of the Hedgehog signaling pathway. A selection of currently known inhibitors, including microRNAs and small molecular inhibitors (both natural and synthetic) are shown, along with their corresponding targets
Currently, most drugs targeted against the Hh pathway function by inhibiting Smo and thus lead to the suppression of tumor proliferation. However, Hh signaling could also be altered by targeting components located downstream of Smo. Accordingly, several groups are attempting to develop agents that target Gli or other molecules in the Hh pathway. For example, GANT61 is an Hh inhibitor targeting Gli1 and Gli2. GANT61 has been shown to effectively down-regulate Gli expression, inhibit cell proliferation and migration, and induce G1 arrest and apoptosis.[69] GANT61 may also decrease cell invasiveness by inhibiting Gli2 in human bladder transitional cell carcinoma.[70] Another potential therapeutic agent is arsenic trioxide (ATO). ATO has been proposed to block the accumulation of Gli2, resulting in reduced protein levels,[71] and to bind directly to Gli1, inhibiting its transcriptional function.[72] Furthermore, four Hh pathway inhibitors (HPIs) have been identified that act downstream of Sufu to modulate Gli processing, activation, and/or trafficking, including small molecule antagonist of ciliogenesis. HPI-1 has been shown to inhibit activation of the Hh pathway induced by overexpression of Gli1. HPI-2, on the other hand, inhibits Hh target gene expression in cells lacking Sufu function or overexpressing Gli2, but is less effective against exogenous Gli1. HPI-3 likely blocks activation by Gli2 as well, albeit through a different mechanism. HPI-4 appears to act by perturbing ciliogenesis.[73] Another synthetic molecule, named Robotnikinin, specifically binds to extracellular Shh and blocks Shh- signaling in cell lines, human primary keratinocytes and a synthetic model of human skin,[74] which may represent an alternative treatment for tumors resistant to Smo inhibitors.
In addition to synthetic drugs, several natural molecules have been found to target the Hh pathway. Genistein is an isoflavone found in soybeans and most soy- protein products. Both in vitro and vivo studies have demonstrated that genistein may inhibit Gli1 mRNA expression and down-regulate Gli reporter activity, leading to significant inhibition of prostate cancer cell proliferation.[75] Cyclopamine, derived from Veratrum californicum, has been showed directly to bind to Smo, blocking the Hh pathway and preventing transcription.[76] In addition, cyclopamine reduces neurosphere formation in glioblastoma cell lines.[77] Epigallocatechin-3-gallate (EGCG) is one of the most potent anticarcinogenic compounds known as catechins found in green tea. Studies indicate that EGCG could decrease the expression of Gli1 mRNA and inhibit Gli reporter activity.[75] Another study found that EGCG down-regulates Ihh, Gli1, and Bcl-2 expression, which may inhibit cell proliferation and induce apoptosis.[78] Another natural inhibitor of the Hh pathway is resveratrol, a stilbenoid found in the skin of red grapes and peanuts. Experimental studies have shown that resveratrol suppresses cancer cell proliferation and induces apoptosis partially through the down-regulation of Gli1 mRNA expression and the inhibition of Gli reporter activity.[75] Curcumin (diferuloylmethane), derived from Curcuma longa, inhibits the nuclear factor-κB, PI3K/Akt, and activator protein-1 signaling pathways, resulting in antiinflammatory, antioxidant and anticancer effects. Curcumin also regulates Hh signaling by down-regulating Shh and Gli1.[79] In addition, curcumin reduces the protein levels of β-catenin and its downstream targets, c-Myc and cyclin D1, suggesting that curcumin could interrupt the cross-talk between Hh and Wnt signaling.[79] In an advanced model of pancreatic cancer, cyclopamine alone was not sufficient to deplete the number of CSCs. Following treatment with a combination of the conventional chemotherapy agent gemcitabine and the mTOR inhibitor rapamycin, however, CSCs were virtually undetectable both in vitro and in vivo.[80] Natural molecules targeting a variety of different pathways involved in cancer proliferation have been identified, and may lead to effective therapies for glioma or other malignancies. It is possible, however, that combination therapy may be needed for treatment of gliomas. Therefore, the exact mechanism of each inhibitor should be investigated, and the effects and defects of combined therapy should be evaluated in clinical trials [Table 3 and Figure 2].
Hh related natural products
| Natural compounds | Target |
|---|---|
| Cyclopamine | Smo |
| Genistein | Gli1 |
| Curcumin (diferuloylmethane) | Shh, Gli1 |
| Epigallocatechin-3-gallate | Ihh, Gli1 |
| Resveratrol | Gli1 |
Conclusion
The Hh signaling pathway plays a pivotal role in the process of embryonic development. Aberrant activation of Hh signaling contributes to cancer development, progression, and the processes of cancer invasion and metastasis, leading to the formation of gliomas. GSCs are at the core of glioma biology and play an important role in cancer invasion, metastasis, drug resistance and tumor recurrence. Thus, strategies specifically targeting Hh signaling in GSCs could lead to promising therapies that inhibit tumor initiation and progression. A variety of synthetic molecules and natural products are currently under investigation in both fundamental research studies and clinical trials. Although some benefits have been observed, there are still problems that remain to be solved. In particular, further studies are needed to (1) identify more effective methods of differentiating GSCs from NSCs; (2) evaluate the benefits of combination therapy with HPIs and conventional chemotherapeutic agents; and (3) determine the mechanisms behind nutraceuticals that inhibit the Hh pathway for the prevention of human malignancies in vitro, in vivo, and in clinical trials.
Acknowledgments
This project was supported by the National High Technology Research and Development Program 863 (2012AA02A508), national 11th 5 years supporting project (2007BA105B08) and Heilongjiang Natural Science key projects (C03030307).
Conflicts of interest
There are no conflicts of interest.
REFERENCES
1. Chen L, Zhang W, Yan W, Han L, Zhang K, Shi Z, Zhang J, Wang Y, Li Y, Yu S, Pu P, Jiang C, Jiang T, Kang C. The putative tumor suppressor miR-524-5p directly targets Jagged-1 and Hes-1 in glioma. Carcinogenesis 2012;33:2276-82.
2. Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO; European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups; National Cancer Institute of Canada Clinical Trials Group. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 2005;352:987-96.
4. Hunter SG, Zhuang G, Brantley-Sieders D, Swat W, Cowan CW, Chen J. Essential role of Vav family guanine nucleotide exchange factors in EphA receptor-mediated angiogenesis. Mol Cell Biol 2006;26:4830-42.
5. L apidot T, Sirard C, Vor moor J, Murdoch B, Hoang T, Caceres-Cortes J, Minden M, Paterson B, Caligiuri MA, Dick JE. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature 1994;367:645-8.
6. Bonnet D, Dick JE. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 1997;3:730-7.
7. Bao S, Wu Q, McLendon RE, Hao Y, Shi Q, Hjelmeland AB, Dewhirst MW, Bigner DD, Rich JN. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 2006;444:756-60.
8. Hambardzumyan D, Squatrito M, Holland EC. Radiation resistance and stem-like cells in brain tumors. Cancer Cell 2006;10:454-6.
9. Gómez-Gaviro MV, Lovell-Badge R, Fer nández-Avilés F, Lara-Pezzi E. The vascular stem cell niche. J Cardiovasc Transl Res 2012;5:618-30.
10. Yoshida S, Sukeno M, Nabeshima Y. A vasculature-associated niche for undifferentiated spermatogonia in the mouse testis. Science 2007;317:1722-6.
11. Gilbertson RJ, Rich JN. Making a tumour's bed: glioblastoma stem cells and the vascular niche. Nat Rev Cancer 2007;7:733-6.
12. Li Z, Bao S, Wu Q, Wang H, Eyler C, Sathornsumetee S, Shi Q, Cao Y, Lathia J, McLendon RE, Hjelmeland AB, Rich JN. Hypoxia-inducible factors regulate tumorigenic capacity of glioma stem cells. Cancer Cell 2009;15:501-13.
13. Wang J, Sakariassen PO, Tsinkalovsky O, Immervoll H, Boe SO, Svendsen A, Prestegarden L, Rosland G, Thorsen F, Stuhr L, Molven A, Bjerkvig R, Enger PO. CD133 negative glioma cells form tumors in nude rats and give rise to CD133 positive cells. Int J Cancer 2008;122:761-8.
14. Reardon DA, Wen PY, Desjardins A, Batchelor TT, Vredenburgh JJ. Glioblastoma multiforme: an emerging paradigm of anti-VEGF therapy. Expert Opin Biol Ther 2008;8:541-53.
15. Jamal M, Rath BH, Tsang PS, Camphausen K, Tofilon PJ. The brain microenvironment preferentially enhances the radioresistance of CD133(+) glioblastoma stem-like cells. Neoplasia 2012;14:150-8.
16. Hardee ME, Marciscano AE, Medina-Ramirez CM, Zagzag D, Narayana A, Lonning SM, Barcellos-Hoff MH. Resistance of glioblastoma-initiating cells to radiation mediated by the tumor microenvironment can be abolished by inhibiting transforming growth factor-β. Cancer Res 2012;72:4119-29.
17. Liu G, Yuan X, Zeng Z, Tunici P, Ng H, Abdulkadir IR, Lu L, Irvin D, Black KL, Yu JS. Analysis of gene expression and chemoresistance of CD133+cancer stem cells in glioblastoma. Mol Cancer 2006;5:67.
18. Piccirillo SG, Reynolds BA, Zanetti N, Lamorte G, Binda E, Broggi G, Brem H, Olivi A, Dimeco F, Vescovi AL. Bone morphogenetic proteins inhibit the tumorigenic potential of human brain tumour-initiating cells. Nature 2006;444:761-5.
19. Kim RK, Yoon CH, Hyun KH, Lee H, An S, Park MJ, Kim MJ, Lee SJ. Role of lymphocyte-specific protein tyrosine kinase (LCK) in the expansion of glioma-initiating cells by fractionated radiation. Biochem Biophys Res Commun 2010;402:631-6.
20. Jamal M, Rath BH, Williams ES, Camphausen K, Tofilon PJ. Microenvironmental regulation of glioblastoma radioresponse. Clin Cancer Res 2010;16:6049-59.
22. Clement V, Sanchez P, de Tribolet N, Radovanovic I, Ruiz i Altaba A. HEDGEHOG-GLI1 signaling regulates human glioma growth, cancer stem cell self-renewal, and tumorigenicity. Curr Biol 2007;17:165-72.
23. Schatton T, Murphy GF, Frank NY, Yamaura K, Waaga-Gasser AM, Gasser M, Zhan Q, Jordan S, Duncan LM, Weishaupt C, Fuhlbrigge RC, Kupper TS, Sayegh MH, Frank MH. Identification of cells initiating human melanomas. Nature 2008;451:345-9.
24. Gunther HS, Schmidt NO, Phillips HS, Kemming D, Kharbanda S, Soriano R, Modrusan Z, Meissner H, Westphal M, Lamszus K. Glioblastoma-derived stem cell-enriched cultures form distinct subgroups according to molecular and phenotypic criteria. Oncogene 2008;27:2897-909.
25. Qiu B, Zhang D, Tao J, Wu A, Wang Y. A simplified and modified procedure to culture brain glioma stem cells from clinical specimens. Oncol Lett 2012;3:50-4.
26. Yu SC, Ping YF, Yi L, Zhou ZH, Chen JH, Yao XH, Gao L, Wang JM, Bian XW. Isolation and characterization of cancer stem cells from a human glioblastoma cell line U87. Cancer Lett 2008;265:124-34.
27. Read TA, Fogarty MP, Markant SL, McLendon RE, Wei Z, Ellison DW, Febbo PG, Wechsler-Reya RJ. Identification of CD15 as a marker for tumor-propagating cells in a mouse model of medulloblastoma. Cancer Cell 2009;15:135-47.
28. Tchoghandjian A, Baeza N, Colin C, Cayre M, Metellus P, Beclin C, Ouafik L, Figarella-Branger D. A2B5 cells from human glioblastoma have cancer stem cell properties. Brain Pathol 2010;20:211-21.
29. Ogden AT, Waziri AE, Lochhead RA. Identification of A2B5+CD133- tumor initiating cells in adult human gliomas. Neurosurgery 2008;62:505-15.
30. Bao S, Wu Q, Li Z, Sathornsumetee S, Wang H, McLendon RE, Hjelmeland AB, Rich JN. Targeting cancer stem cells through L1CAM suppresses glioma growth. Cancer Res 2008;68:6043-8.
31. Ogden AT, Waziri AE, Lochhead RA, Fusco D, Lopez K, Ellis JA, Kang J, Assanah M, McKhann GM, Sisti MB, McCormick PC, Canoll P, Bruce JN. Identification of A2B5+CD133- tumor-initiating cells in adult human gliomas. Neurosurgery 2008;62:505-14.
32. Clément V, Marino D, Cudalbu C, Hamou MF, Mlynarik V, de Tribolet N, Dietrich PY, Gruetter R, Hegi ME, Radovanovic I. Marker-independent identification of glioma-initiating cells. Nat Methods 2010;7:224-8.
33. Dlugosz AA, Talpaz M. Following the Hedgehog to new cancer therapies. N Engl J Med 2009;361:1202-5.
34. Yoo YA, Kang MH, Lee HJ, Kim BH, Park JK, Kim HK, Kim JS, Oh SC. Sonic Hedgehog pathway promotes metastasis and lymphangiogenesis via activation of Akt, EMT, and MMP-9 pathway in gastric cancer. Cancer Res 2011;71:7061-70.
35. Uchida H, Arita K, Yunoue S, Yonezawa H, Shinsato Y, Kawano H, Hirano H, Hanaya R, Tokimura H. Role of sonic Hedgehog signaling in migration of cell lines established from CD133-positive malignant glioma cells. J Neurooncol 2011;104:697-704.
36. Po A, Ferretti E, Miele E, De Smaele E, Paganelli A, Canettieri G, Coni S, Di Marcotullio L, Biffoni M, Massimi L, Di Rocco C, Screpanti I, Gulino A. Hedgehog controls neural stem cells through p53-independent regulation of Nanog. EMBO J 2010;29:2646-58.
37. Hsieh A, Ellsworth R, Hsieh D. Hedgehog/GLI1 regulates IGF dependent malignant behaviors in glioma stem cells. J Cell Physiol 2011;226:1118-27.
38. Ulasov IV, Nandi S, Dey M, Sonabend AM, Lesniak MS. Inhibition of Sonic Hedgehog and Notch pathways enhances sensitivity of CD133(+) glioma stem cells to temozolomide therapy. Mol Med 2011;17:103-12.
39. Takezaki T, Hide T, Takanaga H, Nakamura H, Kuratsu J, Kondo T. Essential role of the Hedgehog signaling pathway in human glioma-initiating cells. Cancer Sci 2011;102:1306-12.
41. Fareh M, Turchi L, Virolle V, Debruyne D, Almairac F, De-La-Forest Divonne S, Paquis P, Preynat-Seauve O, Krause KH, Chneiweiss H, Virolle T. The miR 302-367 cluster drastically affects self-renewal and infiltration properties of glioma-initiating cells through CXCR4 repression and consequent disruption of the SHH-GLI-NANOG network. Cell Death Differ 2012;19:232-44.
42. Sengupta R, Dubuc A, Ward S, Yang L, Northcott P, Woerner BM, Kroll K, Luo J, Taylor MD, Wechsler-Reya RJ, Rubin JB. CXCR4 activation defines a new subgroup of sonic Hedgehog-driven medulloblastoma. Cancer Res 2012;72:122-32.
43. Wu LF, Gao L, Hou XM, Zhang QH, Li S, Yang YF, Lin XH. Drosophila miR-5 suppresses Hedgehog signaling by directly targeting smoothened. FEBS Lett 2012;586:4052-60.
44. Ferretti E, De Smaele E, Miele E, Laneve P, Po A, Pelloni M, Paganelli A, Di Marcotullio L, Caffarelli E, Screpanti I, Bozzoni I, Gulino A. Concerted microRNA control of Hedgehog signalling in cerebellar neuronal progenitor and tumour cells. EMBO J 2008;27:2616-27.
45. Jiang Z, Cushing L, Ai X, Lü J. miR-326 Is downstream of sonic Hedgehog signaling and regulates the expression of Gli2 and smoothened. Am J Respir Cell Mol Biol 2014;51:273-83.
46. Flynt AS, Li N, Thatcher EJ, Solnica-Krezel L, Patton JG. Zebrafish miR-214 modulates Hedgehog signaling to specify muscle cell fate. Nat Genet 2007;39:259-63.
47. Hu HY, He L, Fominykh K, Yan Z, Guo S, Zhang X, Taylor MS, Tang L, Li J, Liu J, Wang W, Yu H, Khaitovich P. Evolution of the human-specific microRNA miR-941. Nat Commun 2012;3:1145.
48. Zhang B, Yang XJ, Yu SP, Ming HL, Chen C, Ren BC, Liu ZF, Liu B. Role of Rac1 in the SDF-1-induced migration and invasion of human glioma cell line U251. Zhonghua Yi Xue Za Zhi 2012;92:727-30.
49. Mott JL, Kurita S, Cazanave SC, Bronk SF, Werneburg NW, Fernandez-Zapico ME. Transcriptional suppression of mir-29b-1/ mir-29a promoter by c-Myc, Hedgehog, and NF-kappaB. J Cell Biochem 2010;110:1155-64.
50. Huelsken J, Birchmeier W. New aspects of Wnt signaling pathways in higher vertebrates. Curr Opin Genet Dev 2001;11:547-53.
51. Kalderon D. Similarities between the Hedgehog and Wnt signaling pathways. Trends Cell Biol 2002;12:523-31.
52. Chen Y, Yue S, Xie L, Pu XH, Jin T, Cheng SY. Dual Phosphorylation of suppressor of fused (Sufu) by PKA and GSK3beta regulates its stability and localization in the primary cilium. J Biol Chem 2011;286:13502-11.
53. He J, Sheng T, Stelter AA, Li C, Zhang X, Sinha M, Luxon BA, Xie J. Suppressing Wnt signaling by the Hedgehog pathway through sFRP-1. J Biol Chem 2006;281:35598-602.
54. Stockhausen MT, Kristoffersen K, Poulsen HS. The functional role of Notch signaling in human gliomas. Neuro Oncol 2010;12:199-211.
55. Androutsellis-Theotokis A, Leker RR, Soldner F, Hoeppner DJ, Ravin R, Poser SW, Rueger MA, Bae SK, Kittappa R, McKay RD. Notch signalling regulates stem cell numbers in vitro and in vivo. Nature 2006;442:823-6.
56. Ingram WJ, McCue KI, Tran TH, Hallahan AR, Wainwright BJ. Sonic Hedgehog regulates Hes1 through a novel mechanism that is independent of canonical Notch pathway signalling. Oncogene 2008;27:1489-500.
57. Zhao X, Malhotra GK, Lele SM, Lele MS, West WW, Eudy JD, Band H, Band V. Telomerase-immortalized human mammary stem/ progenitor cells with ability to self-renew and differentiate. Proc Natl Acad Sci U S A 2010;107:14146-51.
58. Hynes NE, Lane HA. ERBB receptors and cancer: the complexity of targeted inhibitors. Nat Rev Cancer 2005;5:341-54.
59. Laner-Plamberger S, Kaser A, Paulischta M, Hauser-Kronberger C, Eichberger T, Frischauf AM. Cooperation between GLI and JUN enhances transcription of JUN and selected GLI target genes. Oncogene 2009;28:1639-51.
60. Kasper M, Schnidar H, Neill GW, Hanneder M, Klingler S, Blaas L, Schmid C, Hauser-Kronberger C, Regl G, Philpott MP, Aberger F. Selective modulation of Hedgehog/GLI target gene expression by epidermal growth factor signaling in human keratinocytes. Mol Cell Biol 2006;26:6283-98.
61. Bigelow RL, Jen EY, Delehedde M, Chari NS, McDonnell TJ. Sonic Hedgehog induces epidermal growth factor dependent matrix infiltration in HaCaT keratinocytes. J Invest Dermatol 2005;124:457-65.
62. Palma V, Lim DA, Dahmane N, Sanchez P, Brionne TC, Herzberg CD, Gitton Y, Carleton A, Alvarez-Buylla A, Ruiz i Altaba A. Sonic Hedgehog controls stem cell behavior in the postnatal and adult brain. Development 2005;132:335-44.
63. Johnson RW, Nguyen MP, Padalecki SS, Grubbs BG, Merkel AR, Oyajobi BO, Matrisian LM, Mundy GR, Sterling JA. TGF-beta promotion of Gli2-induced expression of parathyroid hormone-related protein, an important osteolytic factor in bone metastasis, is independent of canonical Hedgehog signaling. Cancer Res 2011;71:822-31.
64. Ju B, Spitsbergen J, Eden CJ, Taylor MR, Chen W. Co-activation of Hedgehog and AKT pathways promote tumorigenesis in zebrafish. Mol Cancer 2009;8:40.
65. Rudin CM, Hann CL, Laterra J, Yauch RL, Callahan CA, Fu L, Holcomb T, Stinson J, Gould SE, Coleman B, LoRusso PM, Von Hoff DD, de Sauvage FJ, Low JA. Treatment of medulloblastoma with Hedgehog pathway inhibitor GDC-0449. N Engl J Med 2009;361:1173-8.
66. Dijkgraaf GJ, Alicke B, Weinmann L, Januario T, West K, Modrusan Z, Burdick D, Goldsmith R, Robarge K, Sutherlin D, Scales SJ, Gould SE, Yauch RL, de Sauvage FJ. Small molecule inhibition of GDC-0449 refractory smoothened mutants and downstream mechanisms of drug resistance. Cancer Res 2011;71:435-44.
67. Buonamici S, Williams J, Morrissey M, Wang A, Guo R, Vattay A, Hsiao K, Yuan J, Green J, Ospina B, Yu Q, Ostrom L, Fordjour P, Anderson DL, Monahan JE, Kelleher JF, Peukert S, Pan S, Wu X, Maira SM, Garcia-Echeverria C, Briggs KJ, Watkins DN, Yao YM, Lengauer C, Warmuth M, Sellers WR, Dorsch M. Interfering with resistance to smoothened antagonists by inhibition of the PI3K pathway in medulloblastoma. Sci Transl Med 2010;2:51ra70.
68. Lee MJ, Hatton BA, Villavicencio EH, Khanna PC, Friedman SD, Ditzler S, Pullar B, Robison K, White KF, Tunkey C, LeBlanc M, Randolph-Habecker J, Knoblaugh SE, Hansen S, Richards A, Wainwright BJ, McGovern K, Olson JM. Hedgehog pathway inhibitor saridegib (IPI-926) increases lifespan in a mouse medulloblastoma model. Proc Natl Acad Sci U S A 2012;109:7859-64.
69. Yan M, Wang L, Zuo H, Zhang Z, Chen W, Mao L, Zhang P. HH/ GLI signalling as a new therapeutic target for patients with oral squamous cell carcinoma. Oral Oncol 2011;47:504-9.
70. Mechlin CW, Tanner MJ, Chen M, Buttyan R, Levin RM, Mian BM. Gli2 expression and human bladder transitional carcinoma cell invasiveness. J Urol 2010;184:344-51.
71. Kim J, Lee JJ, Kim J, Gardner D, Beachy PA. Arsenic antagonizes the Hedgehog pathway by preventing ciliary accumulation and reducing stability of the Gli2 transcriptional effector. Proc Natl Acad Sci U S A 2010;107:13432-7.
72. Beauchamp EM, Ringer L, Bulut G, Sajwan KP, Hall MD, Lee YC, Peaceman D, Ozdemirli M, Rodriguez O, Macdonald TJ, Albanese C, Toretsky JA, Uren A. Arsenic trioxide inhibits human cancer cell growth and tumor development in mice by blocking Hedgehog/ GLI pathway. J Clin Invest 2011;121:148-60.
73. Hyman JM, Firestone AJ, Heine VM, Zhao Y, Ocasio CA, Han K, Sun M, Rack PG, Sinha S, Wu JJ, Solow-Cordero DE, Jiang J, Rowitch DH, Chen JK. Small-molecule inhibitors reveal multiple strategies for Hedgehog pathway blockade. Proc Natl Acad Sci U S A 2009;106:14132-7.
74. Stanton BZ, Peng LF, Maloof N, Nakai K, Wang X, Duffner JL, Taveras KM, Hyman JM, Lee SW, Koehler AN, Chen JK, Fox JL, Mandinova A, Schreiber SL. A small molecule that binds Hedgehog and blocks its signaling in human cells. Nat Chem Biol 2009;5:154-6.
75. Slusarz A, Shenouda NS, Sakla MS, Drenkhahn SK, Narula AS, MacDonald RS, Besch-Williford CL, Lubahn DB. Common botanical compounds inhibit the Hedgehog signaling pathway in prostate cancer. Cancer Res 2010;70:3382-90.
76. Chen JK, Taipale J, Cooper MK, Beachy PA. Inhibition of Hedgehog signaling by direct binding of cyclopamine to smoothened. Genes Dev 2002;16:2743-8.
77. Bar EE, Chaudhry A, Lin A, Fan X, Schreck K, Matsui W, Piccirillo S, Vescovi AL, DiMeco F, Olivi A, Eberhart CG. Cyclopamine-mediated Hedgehog pathway inhibition depletes stem-like cancer cells in glioblastoma. Stem Cells 2007;25:2524-33.
78. Tang GQ, Yan TQ, Guo W, Ren TT, Peng CL, Zhao H, Lu XC, Zhao FL, Han X. (-)-Epigallocatechin-3-gallate induces apoptosis and suppresses proliferation by inhibiting the human Indian Hedgehog pathway in human chondrosarcoma cells. J Cancer Res Clin Oncol 2010;136:1179-85.
79. Elamin MH, Shinwari Z, Hendrayani SF, Al-Hindi H, Al-Shail E, Khafaga Y, Al-Kofide A, Aboussekhra A. Curcumin inhibits the Sonic Hedgehog signaling pathway and triggers apoptosis in medulloblastoma cells. Mol Carcinog 2010;49:302-14.
80. Mueller MT, Hermann PC, Witthauer J, Rubio-Viqueira B, Leicht SF, Huber S, Ellwart JW, Mustafa M, Bartenstein P, D'Haese JG, Schoenberg MH, Berger F, Jauch KW, Hidalgo M, Heeschen C. Combined targeted treatment to eliminate tumorigenic cancer stem cells in human pancreatic cancer. Gastroenterology 2009;137:1102-13.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Liu Y, Liu X, Chen LC, Du WZ, Cui YQ, Piao XY, Li YL, Jiang CL. Targeting glioma stem cells via the Hedgehog signaling pathway. Neurosciences 2014;1:51-9. http://dx.doi.org/10.4103/2347-8659.139715
AMA Style
Liu Y, Liu X, Chen LC, Du WZ, Cui YQ, Piao XY, Li YL, Jiang CL. Targeting glioma stem cells via the Hedgehog signaling pathway. Neuroimmunology and Neuroinflammation. 2014; 1: 51-9. http://dx.doi.org/10.4103/2347-8659.139715
Chicago/Turabian Style
Liu, Yang, Xing Liu, Ling-Chao Chen, Wen-Zhong Du, Yu-Qiong Cui, Xing-Yin Piao, Yong-Li Li, Chuan-Lu Jiang. 2014. "Targeting glioma stem cells via the Hedgehog signaling pathway" Neuroimmunology and Neuroinflammation. 1: 51-9. http://dx.doi.org/10.4103/2347-8659.139715
ACS Style
Liu, Y.; Liu X.; Chen L.C.; Du W.Z.; Cui Y.Q.; Piao X.Y.; Li Y.L.; Jiang C.L. Targeting glioma stem cells via the Hedgehog signaling pathway. Neurosciences. 2014, 1, 51-9. http://dx.doi.org/10.4103/2347-8659.139715
About This Article
Copyright
Data & Comments
Data
 Cite This Article 19 clicks
Cite This Article 19 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.