The role of neuroinflammation in juvenile bipolar disorder
Abstract
A pathophysiological relationship has been reported between inflammatory processes, decreased levels of neurotrophins, increased oxidative stress and psychiatric disorders in both juvenile and adult ages. Moreover, this relationship remains unclear in juvenile bipolar disorder (BD). We performed a systematic literature review of studies reporting measurements of inflammatory markers, oxidative stress markers or neurotrophins in juvenile and young adult subjects with BD. Concordant findings showed that inflammatory markers are increased since the earlier stages of BD. A positive correlation between decreased levels of a peripheral brain-derived neurotrophic factor and juvenile BD is controversial suggesting that those changes might occur only during the late stage of BD. No changes in central GSH levels were reported in young adult age BD indicating that oxidative stress may be an outcome of long illness duration and repeated affective episodes. In conclusion, preliminary findings indicate that certain relationship exists between inflammatory process and juvenile BD but evidences are insufficient to support a causal relationship. Adequately powered and prospective studies are warranted to clarify the role of inflammation, neurotrophins and oxidative stress in juvenile BD.
Keywords
Introduction
During the last 20 years, a growing body of evidences has supported a pathophysiological relationship between inflammatory processes, decreased neurotrophins levels, increased oxidative stress and psychiatric disorders in both juvenile and adult ages.[1,2]
Multiple studies analyzing peripheral biomarkers of mood disorders have provided important information on the pathophysiologic process underlying adult bipolar disorder (BD).[1,3] Concordant and consistent evidences have shown that brain-derived neurotrophic factor (BDNF) decreases during both manic and depressive phases of bipolar illness,[4-6] increases after the treatment with antidepressant and antimanics[7-9] and correlate with the illness stage with decreased levels in the late stage of BD.[10]
Several independent laboratories have found that depressive and manic states are associated with an imbalance between peripheral levels of pro- and anti-inflammatory cytokines with proteomic analysis revealing that inflammatory pathways are associated with BD and modified by mood-stabilizing lithium treatment.[1,11] Furthermore, adult subjects with BD are at higher risk of developing comorbid medical illnesses such as diabetes, metabolic and cardiovascular diseases that are also associated with elevated levels of pro-inflammatory markers.[12,13] Potentially involved cytokines include tumor necrosis factor (TNF), interleukin-2 (IL-2), IL-6, IL-8, IL-13 and apolipoprotein A1.[14-18]
Finally, growing evidences are showing that increased levels of oxidative stress may be linked to inflammatory and neuroplasticity pathways[19] and play a role in the pathophysiology of bipolar disorder (BD).[20] A meta-analysis found a significant elevation of oxidative stress biomarkers, such as thiobarbituric acid reactive substances (TBARS) and nitric oxide (NO), during all phases of bipolar illness and preliminary data indicated that oxidative stress may be corrected with pharmacological treatments.[5,6]
As mood disorders have a relatively young median age of onset,[21] in the last 30 years pediatric mood disorders have been studied more systematically, especially depression and BD.[22-24] In addition, studying clinical features of mood disorders at onset in the offspring of adults with depression or BD has become a promising research approach.[25,26]
Several reports have shown a relationship between: (1) the dysregulation of inflammatory markers (increased levels of IL-6, IL-1β, IL-2, IL-10, INF-α and TNF); (2) genetic variation in inflammatory genes [C-reactive protein (CRP)-gene polymorphism] and pediatric major depressive disorder;[16] (3) changes in gene expression among subjects with active mood disorders;[16] (4) preliminary evidences of an association between inflammation and suicidality in depressed youths (decreased TNF-α levels in suicidal compared to nonsuicidal depressed adolescents[27]) as well as increased mRNA and protein expression of IL-1β, IL-6 and TNF-α in Brodmann area 10 of suicide victims relative to controls.[28]
Among children and adolescents with BD, there is a high prevalence of conditions associated with inflammation, such as asthma, cardiovascular disorders, diabetes and obesity,[12] often associated with inflammatory markers,[13] including elevated high-sensitivity-C-reactive protein (hsCRP) and IL-6.[29] This is even more striking considering that subjects with asthma, allergies and other inflammatory conditions were routinely excluded from psychiatric samples.
Furthermore, recent studies have also examined the potential psychiatric applications of anti-inflammatory medications, including aspirin, non-steroidal anti-inflammatory drugs, TNF-α antagonists, and omega-3 fatty acids, in the treatment of mood disorders.[30-34]
Given the increasing interest in the field of neuroinflammatory mechanisms and mood disorders, we carried out a systematic review of literature analyzing the potential pathogenic role of inflammatory processes, decreased neurotrophin levels and oxidative stress in the pathogenesis of juvenile BD.
Search algorithm and inclusion criteria
We performed a literature search through PubMed using the following search algorithm: (bipolar disorder OR mania OR bipolar depression) AND (child* OR adolesc* OR youth) AND (neuroinflamm* OR inflamm* OR neurovascular OR neurotrophin* OR oxidative stress). Reports found through cross-references were also reviewed and added if they met established search criteria.
We included only original studies specifically reporting measurements of inflammatory markers or oxidative stress markers or neurotrophins in subjects diagnosed with BD. We used the following inclusion criteria: (1) original research; (2) diagnosis of BD; (3) measurement of at least one inflammatory marker or neurotrophin or oxidative stress marker; (4) subjects’ age younger than 35 years; (5) reports in English language.
Two psychiatrists screened the article titles for potential relevance, reviewed the identified abstracts and selected the full-text papers potentially meeting the inclusion criteria. The papers not meeting established criteria were excluded.
The following variables were extracted from the reviewed reports: study sample size, type of study, subject age range, subject diagnosis, type of rating scales and diagnostic interviews, measurement method and type of inflammatory marker investigated and main findings of the report.
Main findings
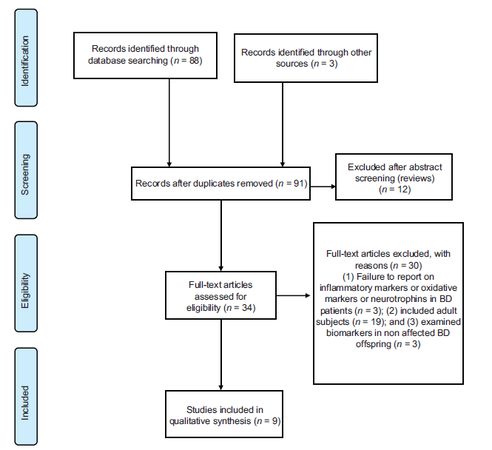
Nine papers were identified through the initial database search and three adjunctive reports were found from cross-references leading to a total of ninety-one screened papers [Figure 1]. Twelve reviews were excluded during the abstract screening. Thirty-four full-text papers were assessed for eligibility and thirty of them were excluded due to either (1) failure to report on inflammatory or oxidative stress markers or neurotrophins (n = 3); (2) included subjects with age > 35 years (n = 19); or (3) included nonaffected bipolar offspring (n = 3). Thus, 9 studies met all our inclusion and exclusion criteria and were included in this review.
Table 1 provides the characteristics of each study, number of included subjects, diagnosis at baseline, age range of the subject sample, considered inflammatory/oxidative stress markers or neurotrophins, and main findings of the considered study.
Summary of studies on biological markers in juvenile bipolar disorder
| Study | Type of study | Subjects (Dx) | Age (years) | Psychiatric measures | Biological markers | Main findings |
|---|---|---|---|---|---|---|
| Barzman et al.[35] | Cross-sectional | n = 10 (BD-I) | 12-17 | WASH-UKSADS; BRACHA | TNF gene | TNF gene expression correlates with both brain activations in amygdala, ACG, and OFC and aggression in adolescents with BD |
| Birmaher et al.[22] | Cross-sectional | n = 30 (18 BD-I; 1 BD-II; 11 BD-NOS) | 12-19 | K-SADS-PL; MRS; FHS | IL-6; hsCRP; BDNF | Manic symptom severity was significantly associated with hsCRP levels. BDNF levels were not correlated with any illness phase |
| Pandey et al.[37] | Longitudinal (8 weeks) | n = 47 (26 BD; 21 HC) | 7-17 | YMRS; CDRS-R | BDNF | Lymphocyte BDNF mRNA and platelet. BDNF levels in drug-free BD subjects were significantly lower than in HCs. Lymphocyte BDNF mRNA was significantly increased in medicated. BD subjects compared to drug-free BD subjects. Lymphocyte BDNF mRNA levels in long-term treated. BD subjects was similar to HCs |
| Chitty et al.[39] | Cross-sectional | n = 50 (24 BD-II; 9 BD-spectrum; 17 HC) | BD 18-30; HC 20-29 | AUDIT; DASS; Kessler-10 | GSH | Decreased GSH in the ACC of high risk drinkers BD subjects. No differences in GSH concentration between BD subjects and HCs |
| Lagopoulos et al.[38] | Cross-sectional | n = 104 (13 BD-I; 25 BD-II; 15 BP-spectrum; 51 HC) | 16-33 | HDRS; BPRS; YMRS; SOFAS; K-10 | GSH | No differences in GSH concentration. between BD subjects and HCs. No significant association between GSH and age of onset or duration of illness. No significant correlations between GSH concentration and mania or depressive symptoms |
| Kauer-Sant’Anna, et al.[10] | Cross-sectional | n = 120 (30 BD-I early-stage; 30 BD-I late-stage; 60 HC) | Early-stage BD 15-35; late-stage BD 18-65 | YMRS; HAMD-21; GAF | BDNF; TNF-α; IL-6; IL-10 | Decreased BDNF levels in late-stage BD patients compared to HCs. Higher TNF-α and IL-6 levels in BD subjects than in HCs during both early and late stage BD. Significant negative correlation between length of illness and decreased BDNF levels. Positive correlation between TNF-α levels and length of illness |
| Magalhaes et al.[40] | Cross-sectional | n = 231 (33 BD-I; 22 BD-II; 82 MDD; 94 HC) | 18-24 | SCID | PCC; TBARS | Higher PCC levels BD subjects than in HCs. No change in TBARS levels between BD subjects and HCs. MDD were not different from control subjects in either PCC or TBARS levels. PCC or TBARS levels could not differentiate MDD from BD subjects. MDD and BD duration of illness did not correlate with either TBARS or PCC. Serum PCC levels were associated with a current manic episode. Serum TBARS levels were not associated with mania or depression |
| Su et al.[36] | Cross-sectional | n = 62 (10 bipolar depression; 13 reactive depression; 18 major depression; 21 HC) | 18-30 | BPRS; HAM-D | BDNF; adiponectin; hsCRP; TNF-α; IL-6 | All depressed groups had serum BDNF levels lower than HCs. No differences in BDNF levels between depressive subtypes. Plasma adiponectin was lower in BD subjects than in HCs. TNF-α was significantly higher in depressed patients than in HCs. No differences in TNF-α levels between depressive subtypes. No differences in IL-6 and hsCRP concentrations were found between depressed and healthy subjects or between depressive subtypes |
| Wiener et al.[41] | Cross-sectional | n = 231 (82 MDD; 33 BD-I; 22 BD-II; 94 HC) | 18-24 | HDRS; YMRS; ASSIST | Uric acid; PCC; TBARS | No association between oxidative stress parameters and clinical diagnosis of MDD and BD for women and men |
Pro-inflammatory markers
One study examined serum pro-inflammatory markers IL-6 and hsCRP and serum BDNF among 30 adolescents diagnosed with BD [18 bipolar type I disorder (BD-I), 1 bipolar type II disorder (BD-II) and 11 BD not otherwise specified] from the Course and Outcome Bipolar Youth study.[29] They found a positive association between manic and hypomanic symptom severity and hsCRP levels. Manic symptom severity was associated with high levels of hsCRP, but not with IL-6 serum levels. Notably all 3 subjects with hsCRP levels > 10 μg/mL had a very high manic symptom score (Mania Rating Scale, MRS > 20). Depressive symptom severity was not significantly associated with hsCRP or IL-6 serum levels. Forty percent of participants had levels of hsCRP that are considered at risk for cardiovascular diseases among adults.
Barzman et al.[35] examined the associations between TNF gene expressions, functional brain activation under a frustrative non-reward task and aggression in a sample of 10 adolescents affected by BD-I. They found that gene expression of protein in the TNF pathways correlates with both activation in amygdala, anterior cingulate cortex (ACC) and orbito-frontal cortex and aggression in adolescents with BD suggesting that TNF-related inflammatory genes may play a role in neural activity associated with frustrative non-reward and aggressive behaviors in pediatric BD.
Su et al.[36] investigated pro-inflammatory cytokines levels in a cohort of young males suffering from reactive depression or major depression, or bipolar depression compared to matched sample of healthy control subjects. They found significantly higher levels of TNF-α and significantly lower levels of adiponectin in depressed youths compared to healthy controls, with no difference in both TNF-α and adiponectin levels between depressive subtypes.[36] No difference was found in IL-6 and hsCRP levels between depressed and healthy subjects and between different subtypes of depression.[36] Consistently with these findings supporting early changes in pro-inflammatory cytokine levels during the psychopathological development of BD, Kauer-Sant’Anna et al.[10] found that TNF-α and IL-6 levels were already significantly increased in early-stage BD patients compared to healthy controls and continued to be higher in BD subjects than controls also in the late-stage of the disease. Additionally, they found a positive correlation between TNF-α levels and length of illness.[10] Conversely, the anti-inflammatory IL-10 levels were increased in the early stage of BD but not in the late stage of BD.[10]
BDNF
Pandey et al.[37] compared gene expression and protein levels of BDNF in a sample of 26 manic or mixed BD adolescents before and after mood-stabilizing treatment with a sample of 21 matched healthy controls. They measured BDNF mRNA levels in lymphocytes of BD subjects before and after treatment and in healthy controls and BDNF protein levels in platelets of drug free BD and healthy subjects. They found that (1) BDNF mRNA levels in lymphocytes and BDNF protein levels in platelets of drug-free subjects with BD were significantly lower compared to those of healthy controls; (2) long-term treatment with mood-stabilizing drugs significantly increased the levels of BDNF mRNA in the lymphocytes of subjects with BD; and that (3) BDNF mRNA level of BD patients during the eighth week of treatment was comparable to that of healthy control subjects.[37]
Measurements of BDNF peripheral levels in a sample of young adult males diagnosed with bipolar depression showed that BDNF levels were significantly lower in depressed subjects than in healthy controls.[36]
These finding were not replicated in a later study[29] reporting that BDNF levels in a sample of BD adolescents were not correlated with any illness phase (depressive or manic), but was significantly and inversely associated with IL-6 levels. Consistently with this last observation, Kauer-Sant’Anna et al.[10] found that BDNF levels were similar between patients with early stage BD and matched controls but were significantly decreased in patients with late-stage BD. The decrease in BDNF levels appeared to be proportional to the length of illness and BDNF levels were negatively correlated to the number of mood episodes.[10]
Oxidative stress
Two studies about the measurement of glutathione (GSH) concentrations in young adult patients with BDcompared to healthy subjects suggested that there wasno difference in GSH level in the ACC between patients and controls.[38,39] Theyreported that GSH levels were not correlated to depressive and manic episode severity[38] and were not significantly different between unmedicated and medicated subjects.[39] Also, they found that GSH levels were decreased in bipolar subjects with high levels of alcohol intake.[39]
Magalhaes et al.[40] suggested that young adults with alifetime history of hypomania had higher levels of oxidative damage to proteins as measured by the determinationof carbonyl groups [protein carbonyl content (PCC)] when compared to healthy young adults. High serum PCC levels were associated with a current manic episode, but not with a current depressive episode. Conversely, the levels of lipid peroxidation as measured using the TBARS method did not significantly differ between mood disorder subjects and healthy controls and did not correlate with manic or depressive mood state.[40]
A significant gender-related difference in oxidative stress parameters was reported by the same group[41] showing higher PCC and lower uric acid levels in females when compared to males. No association was found between oxidative stress parameters and bipolar versus major depressive disorder in both genders.[41]
Discussion
The study of inflammatory factors in chronic psychiatric conditions is a relatively new field of research that has already highlighted several important areas of focus in populations of adult bipolar disorder patients.[1,3]
Our review provides a summary of preliminary findings about the link between inflammatory processes, decreased neurotrophins, increased oxidative stress and juvenile or young adult age BD. Two different lines of research have been pursued in this field, one regarding early onset (pediatric) of BD and the other on the effects of course variables (duration of illness, number of episodes, hospitalizations) on changes in inflammatory markers, neurotrophins and markers of oxidative stress. Studying inflammatory mechanisms in pediatric BD could help to understand the relationship between inflammation and mood episodes. This relationship can be causal (thus preceding and predicting the development of a mood disorder), merely associated with the disease or a consequence of a long lasting illness.
Only three studies[29,35,37] examined BDNF and inflammatory markers in small populations of pediatric bipolar disorder, thus the reported findings are mostly preliminary and not replicated. Six additional studies examined the role of oxidative stress, inflammatory cytokines and BDNF in the pathophysiology of early onset bipolar disorder during adult age.[10,36,38-41]
Concordant findings showed that inflammatory markers are increased since the earlier stages of BD with: (1) increased TNF-α gene expression in adolescent BD showing aggressive behaviors;[35] (2) increased TNF-α levels in young adults with bipolar depression;[36] (3) increased TNF-α levels since the earlier stages of BD;[10] and (4) positive correlations between TNF-α levels and length of bipolar illness.[10] Also, increased levels of hsCRP have been detected in juvenile BD patients during manic and mixed episodes.[29]
A positive correlation was found between decreased levels of peripheral BDNF and a manic, depressive or mixed episode in juvenile and young adult BD,[36,37] even though such findings have not been always replicated.[10,29] In fact, some authors have suggested that changes in peripheral levels of BDNF might occur only during the late stage of BD and might reflect the neurodegeneration of late stage mood disorders.[10] Indeed, recent pre-clinical and clinical evidences suggested that the excitoxicity due to an excessive glutamatergic transmission might play a role in the pathogenesis of the hypothesized neurodegeneration associated with BD.[42-44] Also, recent studies have shown that TNF-α is a key cytokine stimulating extensive release of glutamate from microglial cells,[45] while the neuroprotective effect of the mood stabilizing treatments like lithium[46] and the recently suggested promising memantine[47,48] is well known.
Finally, findings examining the role of oxidative stress in juvenile BD are substantially controversial as no changes in central GSH levels was measured in vivo using magnetic resonance spectroscopy during manic or depressive phase of young adult BD.[38,39] These findings are inconsistent with studies from other groups finding increased oxidative stress in older samples with illness duration of 10 years on average[20] indicating that oxidative stress may be an outcome of long illness duration and repeated affective episodes rather than being a core feature of the pathophysiology of BD at onset.
Reasons of weakness and inconsistency across the studies are diverse and include heterogeneity of the samples (age and considered BD phases, concurrent use of drugs, substance abuse, comorbidity with other medical illnesses, effect of other psychiatry conditions, especially anxiety related disorders), small to modest sample sizes and differences in studied biological pathways. Also, it is worth to underscore that peripheral change in biological markers might not always correspond to comparable changes of the same markers in the central nervous system.
Conclusion
There are preliminary findings indicating that potential relationship exists between inflammatory process and juvenile BD, but evidences are insufficient to support the causality. Adequately powered and prospective studies on high risk population as well as studies examining the relationship between mood-stabilizing treatment and changes in inflammatory, oxidative markers and neurotrophins levels are warranted to understand their role in the pathogenesis of BD.
Financial support and sponsorship
It was supported by the Research Fellowship from Sapienza University of Rome to Dr. Giulia Serra.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
1. Frey BN, Andreazza AC, Houenou J, Jamain S, Goldstein BI, Frye MA, Leboyer M, Berk M, Malhi GS, Lopez-Jaramillo C, Taylor VH, Dodd S, Frangou S, Hall GB, Fernandes BS, Kauer-Sant'Anna M, Yatham LN, Kapczinski F, Young LT. Biomarkers in bipolar disorder: a positional paper from the International Society for Bipolar Disorders Biomarkers Task Force. Aust N Z J Psychiatry 2013;47:321-32.
2. Mitchell RH, Goldstein BI. Inflammation in children and adolescents with neuropsychiatric disorders: a systematic review. J Am Acad Child Adolesc Psychiatry 2014;53:274-96.
3. Goldstein BI, Young LT. Toward clinically applicable biomarkers in bipolar disorder: focus on BDNF, inflammatory markers, and endothelial function. Curr Psychiatry Rep 2013;15:425.
4. Cunha AB, Frey BN, Andreazza AC, Goi JD, Rosa AR, Goncalves CA, Santin A, Kapczinski F. Serum brain-derived neurotrophic factor is decreased in bipolar disorder during depressive and manic episodes. Neurosci Lett 2006;398:215-9.
5. Machado-Vieira R, Dietrich MO, Leke R, Cereser VH, Zanatto V, Kapczinski F, Souza DO, Portela LV, Gentil V. Decreased plasma brain derived neurotrophic factor levels in unmedicated bipolar patients during manic episode. Biol Psychiatry 2007;61:142-4.
6. Frey BN, Andreazza AC, Kunz M, Gomes FA, Quevedo J, Salvador M, Goncalves CA, Kapczinski F. Increased oxidative stress and DNA damage in bipolar disorder: a twin-case report. Prog Neuropsychopharmacol Biol Psychiatry 2007;31:283-5.
7. Gonul AS, Akdeniz F, Taneli F, Donat O, Eker C, Vahip S. Effect of treatment on serum brain-derived neurotrophic factor levels in depressed patients. Eur Arch Psychiatry Clin Neurosci 2005;255:381-6.
8. de Sousa RT, van de Bilt MT, Diniz BS, Ladeira RB, Portela LV, Souza DO, Forlenza OV, Gattaz WF, Machado-Vieira R. Lithium increases plasma brain-derived neurotrophic factor in acute bipolar mania: a preliminary 4-week study. Neurosci Lett 2011;494:54-6.
9. Tramontina JF, Andreazza AC, Kauer-Sant'anna M, Stertz L, Goi J, Chiarani F, Kapczinski F. Brain-derived neurotrophic factor serum levels before and after treatment for acute mania. Neurosci Lett 2009;452:111-3.
10. Kauer-Sant'Anna M, Kapczinski F, Andreazza AC, Bond DJ, Lam RW, Young LT, Yatham LN. Brain-derived neurotrophic factor and inflammatory markers in patients with early- vs. late-stage bipolar disorder. Int J Neuropsychopharmacol 2009;12:447-58.
11. Sussulini A, Dihazi H, Banzato CE, Arruda MA, Stuhmer W, Ehrenreich H, Jahn O, Kratzin HD. Apolipoprotein A-I as a candidate serum marker for the response to lithium treatment in bipolar disorder. Proteomics 2011;11:261-9.
12. Herberth M, Koethe D, Levin Y, Schwarz E, Krzyszton ND, Schoeffmann S, Ruh H, Rahmoune H, Kranaster L, Schoenborn T, Leweke MF, Guest PC, Bahn S. Peripheral profiling analysis for bipolar disorder reveals markers associated with reduced cell survival. Proteomics 2011;11:94-105.
13. Brietzke E, Stertz L, Fernandes BS, Kauer-Sant'anna M, Mascarenhas M, Escosteguy Vargas A, Chies JA, Kapczinski F. Comparison of cytokine levels in depressed, manic and euthymic patients with bipolar disorder. J Affect Disord 2009;116:214-7.
14. Kim JW, Szigethy EM, Melhem NM, Saghafi EM, Brent DA. Inflammatory markers and the pathogenesis of pediatric depression and suicide: a systematic review of the literature. J Clin Psychiatry 2014;75:1242-53.
15. Kim YK, Jung HG, Myint AM, Kim H, Park SH. Imbalance between pro-inflammatory and anti-inflammatory cytokines in bipolar disorder. J Affect Disord 2007;104:91-5.
16. O'Brien SM, Scully P, Scott LV, Dinan TG. Cytokine profiles in bipolar affective disorder: focus on acutely ill patients. J Affect Disord 2006;90:263-7.
17. Weiner M, Warren L, Fiedorowicz JG. Cardiovascular morbidity and mortality in bipolar disorder. Ann Clin Psychiatry 2011;23:40-7.
18. Leboyer M, Soreca I, Scott J, Frye M, Henry C, Tamouza R, Kupfer DJ. Can bipolar disorder be viewed as a multi-system inflammatory disease? J Affect Disord 2012;141:1-10.
19. de Gonzalo-Calvo D, Neitzert K, Fernandez M, Vega-Naredo I, Caballero B, Garcia-Macia M, Suarez FM, Rodriguez-Colunga MJ, Solano JJ, Coto-Montes A. Differential inflammatory responses in aging and disease: TNF-alpha and IL-6 as possible biomarkers. Free Radic Biol Med 2010;49:733-7.
20. Berk M, Kapczinski F, Andreazza AC, Dean OM, Giorlando F, Maes M, Yucel M, Gama CS, Dodd S, Dean B, Magalhaes PV, Amminger P, McGorry P, Malhi GS. Pathways underlying neuroprogression in bipolar disorder: focus on inflammation, oxidative stress and neurotrophic factors. Neurosci Biobehav Rev 2011;35:804-17.
21. Merikangas KR, Cui L, Kattan G, Carlson GA, Youngstrom EA, Angst J. Mania with and without depression in a community sample of US adolescents. Arch Gen Psychiatry 2012;69:943-51.
22. Birmaher B, Axelson D, Goldstein B, Strober M, Gill MK, Hunt J, Houck P, Ha W, Iyengar S, Kim E, Yen S, Hower H, Esposito-Smythers C, Goldstein T, Ryan N, Keller M. Four-year longitudinal course of children and adolescents with bipolar spectrum disorders: the Course and Outcome of Bipolar Youth (COBY) study. Am J Psychiatry 2009;166:795-804.
23. Uchida M, Serra G, Zayas L, Kenworthy T, Hughes B, Koster A, Faraone SV, Biederman J. Can manic switches be predicted in pediatric major depression? A systematic literature review. J Affect Disord 2014;172C:300-6.
24. Uchida M, Serra G, Zayas L, Kenworthy T, Faraone SV, Biederman J. Can unipolar and bipolar pediatric major depression be differentiated from each other? A systematic review of cross-sectional studies examining differences in unipolar and bipolar depression. J Affect Disord 2015;176:1-7.
25. Duffy A. The early natural history of bipolar disorder: what we have learned from longitudinal high-risk research. Can J Psychiatry 2010;55:477-85.
26. Mesman E, Hillegers MH, Ambree O, Arolt V, Nolen WA, Drexhage HA. Monocyte activation, brain-derived neurotrophic factor (BDNF), and S100B in bipolar offspring: a follow-up study from adolescence into adulthood. Bipolar Disord 2015;17:39-49.
27. Gabbay V, Klein RG, Alonso CM, Babb JS, Nishawala M, De Jesus G, Hirsch GS, Hottinger-Blanc PM, Gonzalez CJ. Immune system dysregulation in adolescent major depressive disorder. J Affect Disord 2009;115:177-82.
28. Pandey GN, Rizavi HS, Ren X, Fareed J, Hoppensteadt DA, Roberts RC, Conley RR, Dwivedi Y. Proinflammatory cytokines in the prefrontal cortex of teenage suicide victims. J Psychiatr Res 2012;46:57-63.
29. Goldstein BI, Collinger KA, Lotrich F, Marsland AL, Gill MK, Axelson DA, Birmaher B. Preliminary findings regarding proinflammatory markers and brain-derived neurotrophic factor among adolescents with bipolar spectrum disorders. J Child Adolesc Psychopharmacol 2011;21:479-84.
30. Mendlewicz J, Kriwin P, Oswald P, Souery D, Alboni S, Brunello N. Shortened onset of action of antidepressants in major depression using acetylsalicylic acid augmentation: a pilot open-label study. Int Clin Psychopharmacol 2006;21:227-31.
31. Muller N, Schwarz MJ, Dehning S, Douhe A, Cerovecki A, Goldstein-Muller B, Spellmann I, Hetzel G, Maino K, Kleindienst N, Moller HJ, Arolt V, Riedel M. The cyclooxygenase-2 inhibitor celecoxib has therapeutic effects in major depression: results of a double-blind, randomized, placebo controlled, add-on pilot study to reboxetine. Mol Psychiatry 2006;11:680-4.
32. Nery FG, Monkul ES, Hatch JP, Fonseca M, Zunta-Soares GB, Frey BN, Bowden CL, Soares JC. Celecoxib as an adjunct in the treatment of depressive or mixed episodes of bipolar disorder: a double-blind, randomized, placebo-controlled study. Hum Psychopharmacol 2008;23:87-94.
33. Raison CL, Rutherford RE, Woolwine BJ, Shuo C, Schettler P, Drake DF, Haroon E, Miller AH. A randomized controlled trial of the tumor necrosis factor antagonist infliximab for treatment-resistant depression: the role of baseline inflammatory biomarkers. JAMA Psychiatry 2013;70:31-41.
34. Freeman MP, Hibbeln JR, Wisner KL, Davis JM, Mischoulon D, Peet M, Keck PE Jr, Marangell LB, Richardson AJ, Lake J, Stoll AL. Omega-3 fatty acids: evidence basis for treatment and future research in psychiatry. J Clin Psychiatry 2006;67:1954-67.
35. Barzman D, Eliassen J, McNamara R, Abonia P, Mossman D, Durling M, Adler C, DelBello M, Lin PI. Correlations of inflammatory gene pathways, corticolimbic functional activities, and aggression in pediatric bipolar disorder: a preliminary study. Psychiatry Res 2014;224:107-11.
36. Su SC, Sun MT, Wen MJ, Lin CJ, Chen YC, Hung YJ. Brain-derived neurotrophic factor, adiponectin, and proinflammatory markers in various subtypes of depression in young men. Int J Psychiatry Med 2011;42:211-26.
37. Pandey GN, Rizavi HS, Dwivedi Y, Pavuluri MN. Brain-derived neurotrophic factor gene expression in pediatric bipolar disorder: effects of treatment and clinical response. J Am Acad Child Adolesc Psychiatry 2008;47:1077-85.
38. Lagopoulos J, Hermens DF, Tobias-Webb J, Duffy S, Naismith SL, White D, Scott E, Hickie IB. In vivo glutathione levels in young persons with bipolar disorder: a magnetic resonance spectroscopy study. J Psychiatr Res 2013;47:412-7.
39. Chitty KM, Lagopoulos J, Hickie IB, Hermens DF. Risky alcohol use in young persons with emerging bipolar disorder is associated with increased oxidative stress. J Affect Disord 2013;150:1238-41.
40. Magalhaes PV, Jansen K, Pinheiro RT, Colpo GD, da Motta LL, Klamt F, da Silva RA, Kapczinski F. Peripheral oxidative damage in early-stage mood disorders: a nested population-based case-control study. Int J Neuropsychopharmacol 2012;15:1043-50.
41. Wiener C, Rassier GT, Kaster MP, Jansen K, Pinheiro RT, Klamt F, Magalhaes PV, Kapczinski F, Ghisleni G, da Silva RA. Gender-based differences in oxidative stress parameters do not underlie the differences in mood disorders susceptibility between sexes. Eur Psychiatry 2014;29:58-63.
42. Ongur D, Jensen JE, Prescot AP, Stork C, Lundy M, Cohen BM, Renshaw PF. Abnormal glutamatergic neurotransmission and neuronal-glial interactions in acute mania. Biol Psychiatry 2008;64:718-26.
43. Atmaca M, Yildirim H. Altered neurochemical ingredient of hippocampus in patients with bipolar depression. Depress Res Treat 2012;2012:485249.
44. Tanovic A, Alfaro V. Glutamate-related excitotoxicity neuroprotection with memantine, an uncompetitive antagonist of NMDA-glutamate receptor, in Alzheimer's disease and vascular dementia. Rev Neurol 2006;42:607-16.
45. Takeuchi H, Jin S, Wang J, Zhang G, Kawanokuchi J, Kuno R, Sonobe Y, Mizuno T, Suzumura A. Tumor necrosis factor-alpha induces neurotoxicity via glutamate release from hemichannels of activated microglia in an autocrine manner. J Biol Chem 2006;281:21362-8.
46. Gray JD, McEwen BS. Lithium's role in neural plasticity and its implications for mood disorders. Acta Psychiatr Scand 2013;128:347-61.
47. Serra G, Koukopoulos A, De Chiara L, Koukopoulos AE, Tondo L, Girardi P, Baldessarini RJ, Serra G. Three-year, naturalistic, mirror-image assessment of adding memantine to the treatment of 30 treatment-resistant patients with bipolar disorder. J Clin Psychiatry 2015;76:e91-7.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Serra G, Chiara LD, Marangoni C, Faedda GL. The role of neuroinflammation in juvenile bipolar disorder. Neurosciences 2015;2:244-51. http://dx.doi.org/10.4103/2347-8659.167303
AMA Style
Serra G, Chiara LD, Marangoni C, Faedda GL. The role of neuroinflammation in juvenile bipolar disorder. Neuroimmunology and Neuroinflammation. 2015; 2: 244-51. http://dx.doi.org/10.4103/2347-8659.167303
Chicago/Turabian Style
Serra, Giulia, Lavinia De Chiara, Ciro Marangoni, Gianni L. Faedda. 2015. "The role of neuroinflammation in juvenile bipolar disorder" Neuroimmunology and Neuroinflammation. 2: 244-51. http://dx.doi.org/10.4103/2347-8659.167303
ACS Style
Serra, G.; Chiara LD.; Marangoni C.; Faedda GL. The role of neuroinflammation in juvenile bipolar disorder. Neurosciences. 2015, 2, 244-51. http://dx.doi.org/10.4103/2347-8659.167303
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 2 clicks
Cite This Article 2 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.