Interleukin-1β-induced inflammatory signaling in C20 human microglial cells
Abstract
Aim: Increased inflammatory signaling in microglia is implicated in the pathogenesis of neurodegenerative diseases, trauma, psychiatric disorders, and anxiety/depression. Understanding inflammatory signaling in microglia is critical for advancing treatment options. Studying rodent-derived microglia has yielded substantial information, yet, much remains to better understand inflammatory signaling in human microglia. Hence, there is great interest in developing immortalized human microglial cell lines. The C20 human microglial cell line was recently developed and our primary objective was to advance our knowledge of inflammatory signaling in these cells.
Methods: Expression of the microglia specific marker transmembrane protein 119 (TMEM119) was assessed by western blot analysis. Lipopolysaccharide (LPS)- and interleukin-1β (IL-1β)-induced cytokine/chemokine expression was determined by ELISA. Phosphorylation of inhibitory kappa B alpha (IκBα), nuclear factor (NF)-κB p65, and p38 mitogen-activated protein kinase (p38 MAPK) was measured by western blot analysis.
Results: TMEM119 was expressed in unstimulated C20 cells, and to a greater extent in IL-1β-stimulated cells. IL-1β significantly induced IL-6, monocyte chemoattractant protein-1/CCL2, and interferon-γ inducible protein 10/CXCL10 expression. LPS induced CCL2 expression, but not IL-6 or CXCL10 expression. IL-1β induced inflammatory signaling as indicated by increased phosphorylation of IκBα, NF-κB p65 and p38 MAPK.
Conclusion: We provide the first evidence that C20 microglia express TMEM119. This is the initial report of IL-1β-induced activation of IκBα, NF-κB p65, and p38 MAPK and subsequent CXCL10, CCL2 and IL-6 secretion in C20 cells. These findings advance our understanding of inflammatory signaling in C20 cells and support the value of this cell line as a research tool.
Keywords
Introduction
Microglia are resident macrophages in the central nervous system (CNS) and are essential to brain physiology; but also, they are instrumental in response to injury and infection in the CNS (See Wolf et. al.[1] for review). Microglia constantly survey their local environment and respond to extracellular cues (e.g., ATP) to maintain homeostasis[2,3]. Other specific physiological functions of microglia include removal of dead neurons and cellular debris[2,3], synaptic pruning[4], and regulation of synaptic connectivity and plasticity[5-7]. Microglia are also integral to innate immunity and are instrumental in neuroinflammation[8,9]. For instance, activated microglia release of pro-inflammatory mediators including, cytokines [e.g., interleukin (IL)-1β, IL-6][10,11], chemokines (e.g., monocyte chemoattractant protein-1/CCL2, interferon-γ inducible protein 10/CXCL10)[11-13] and reactive oxygen species[14,15]. Microglia also modulate the inflammatory response by releasing anti-inflammatory cytokines such as IL-10[16] and transforming growth factor (TGF)-β[17]. Controlled neuroinflammation is neuroprotective[18], however, excessive or chronic neuroinflammation is neurotoxic, it contributes to neurodegeneration, and disrupts neuronal function[1]. For instance, microglial activation and neuroinflammation are present in neurodegenerative diseases, CNS infection and trauma, as well as psychiatric disorders. Indeed, emerging evidence suggests pharmacological modulation of microglia may be beneficial in treating certain CNS disorders[19-22].
Much has been discovered about microglia function, including inflammatory signaling, using in vitro approaches with primary cell cultures and transformed cell lines[23-25]. Significant insights have been obtained about inflammatory signaling using either primary rat or mouse microglia[26-29] or transformed cell lines such as BV-2 murine microglial cells[26,28-30]. While many of the findings have been observed in primary human microglia[14,31,32], not surprisingly, there have been differences observed[24]. For example, bacterial lipopolysaccharide (LPS) and interferon-γ (IFNγ) are potent inducers of inflammatory signaling (e.g., cytokine/chemokine expression) in both mouse and human microglia[29,32-34], whereas, IL-1β activates human, but not mouse microglia[10,11,35,36]. The lack of responsiveness of mouse microglia to IL-1β stimulation is likely a consequence of very low IL-1 receptor (IL-1R) expression[36].
Therefore, while there has been a wealth of knowledge obtained about rodent-derived microglia, it remains critical to better understand inflammatory signaling in human microglia. Primary human microglia (fetal and adult) remain a necessary tool, but they are relatively difficult and expensive to obtain and use, thus, only a limited number of research groups have access, funds, and expertise to extensively use this approach. Primary human microglia are commercially available (e.g., ScienCell Research Laboratories, cat. #HM-1900) however, they are often in limited supply or not in stock. Therefore, there is increasing interest in immortalized human microglial cell lines as sustainable tools for studying microglia function[37-39]. The importance of human microglial cell lines extends beyond basic understanding of microglia function but also these cell lines serve as a platform for investigating the effects of pharmacologic agents on microglia, with an eye toward drug development.
Recently, a novel, immortalized human microglial cell line (C20) was introduced to the field; the cells maintain microglial morphology, express multiple cell surface microglia markers, and express proinflammatory cytokines following stimulation with tumor necrosis factor-α (TNFα)[37]. We are particularly interested in IL-1β-induced inflammatory signaling in microglia due to the established role of IL-1β in neuroinflammation[40-43]. Interestingly, most studies utilize LPS and/or IFNγ to stimulate microglia in vitro; and to a large extent this is because most in vitro studies utilize rodent cells and these are the stimuli to which they respond. Therefore, the primary objective of our investigation was to advance our knowledge of IL-1β-induced inflammatory signaling in human microglia using C20 human microglial cells.
Methods
Cells
C20 human microglial cells were obtained from David Alvarez-Carbonell, PhD (Case Western Reserve University) and details pertaining to the generation of this cell line were recently reported[37]. Briefly, these investigators obtained human microglia from ScienCell Research Laboratories, Carlsbad, CA (Cat# HM1900) and then immortalized the cells using simian virus 40 large T antigen and hTERT (to facilitate expression of human telomerase reverse transcriptase)[37]. The C20 cells that we obtained were confirmed to be of human origin by the Human Identity Testing Laboratory at Oklahoma State University Center for Health Sciences, which utilized the PowerPlex® 21 System (Promega, Madison, WI), a multiplex short tandem repeat system for human identification, as previously described[44]. For our experiments, cells were used at passages 5-10 and were either seeded in 24-well plates (1 × 105 cells/well) or in 100 mm dishes (3 × 106 cells) depending on the experiment and cultured in growth medium [Dulbecco’s Modified Eagle Medium/Ham’s F-12 50/50 mix supplemented with 2.5 mmol/L L-glutamine (Corning 10-090-CV), 10% fetal bovine serum (Atlanta Biologicals S11550), and 1% penicillin/streptomycin (Lonza 17603E)] until 90% confluent (4-5 days). Medium was replaced with serum-free medium (SFM) 24 h prior to stimulation. Normal human astrocytes (NHA, ScienCell, #HA1800) were maintained as previously described[45].
Stimulus
C20 were stimulated in SFM containing either LPS (E. coli K12, 1 µg/mL; InvivoGen, San Diego, CA) or human recombinant IL-1β (20 ng/mL; Peprotech, Rocky Hill, NJ) for 10 min - 24 h depending on the specific experiment; whereas, NHA were stimulated with IL-1β (3 ng/mL) for 24 h in the single study in which they were used. Details regarding the number of independent experiments and replicate treatments within each experimental run are provided in the figure legends.
Expression of microglial marker
While the precise function of transmembrane protein 119 (TMEM119) has yet to be determined, it is increasingly recognized as a reliable marker of human microglia that discriminates microglia in the brain from blood-derived macrophages[46-49]. We assessed TMEM119 expression by western blot analysis and fluorescent immunocytochemistry. For western blot analysis, whole cell lysates were collected from unstimulated and IL-1β-stimulated C20 cells (cultured in 100 mm dishes), using Triton X-100 lysis buffer (50 mmol/L Tris-HCl, 150 mmol/L NaCl, 10% glycerol, 1% Triton X-100) containing MS-SAFE protease/phosphatase inhibitor (Sigma-Aldrich). Briefly, cells were rinsed with cold phosphate buffered saline (PBS), then lysed in 300 µL of lysis buffer and collected into 1.5 mL tubes. The lysates were then incubated on ice for 45 min with intermittent mixing by inversion. Lysates were centrifuged for 10 min at 20,800 × g and 4 °C. The supernatant, containing whole cell protein was then collected and stored at -80 °C. Thirty micrograms of total protein were loaded on 7.5% polyacrylamide gels (BioRad TGX FastCast Acrylamide kit, #161-0171), electrophoresed and then transferred to polyvinylidene difluoride (PVDF) membrane. Membranes were then incubated at 4 °C for 15-18 h with rocking. Membranes were blocked with 5% bovine serum albumin (BSA) for 2 h prior to incubation with antibodies. Primary antibodies included, anti-TMEM119 (1:100, Sigma #HPA051870), anti-glial fibrillary acidic protein (GFAP; 1:5000, Millipore #MAB360), and anti-β-tubulin (1:1000, Cell Signaling #2146S). Anti-rabbit IgG, AP-linked (1:1000, Cell Signaling #7054S) was used as the secondary antibody. Whole cell lysates from NHA were used for comparison. Restore western blot stripping buffer (Thermo Scientific #21059) was used to remove antibodies and allow for re-labelling of membranes. The blots were scanned in a phosphoimager Typhoon 9410 (GE Healthcare, Uppsala, Sweden) using Amersham ECF Substrate #RPN-5785, and Image J (National Institutes of Health) was used for densitometric analysis.
TMEM119 expression was also assessed by fluorescent immunocytochemistry. Briefly, C20 cells were seeded on glass coverslips in 6-well dishes and cultured as described above until 70%-80% confluent. Following the treatment period (24 h), cells were washed three times with PBS then fixed in 4% paraformaldehyde for 15 min. Cells were washed with PBS and then incubated in blocking buffer (1% BSA in PBS) overnight at room temperature. Cells were then incubated in anti-TMEM119 (1:500 in PBS containing 0.5% BSA, Sigma #HPA051870) overnight at 4 °C with rocking. After washing with PBS, cells were incubated for 2 h in Alexa Fluor® 488 donkey anti-rabbit antibody (1:3000 in PBS containing 0.5% BSA; Life Technologies #A-21206). Cells were washed again with PBS, treated with 300 nmol/L 4ʹ,6-diamidino-2-phenylindole (DAPI; Sigma) without rocking for 15 min at room temperature, then washed in PBS prior to mounting in Prolong Gold anti-fade reagent (Invitrogen; Eugene, OR).
Cytokine/chemokine expression
Levels of secreted CXCL10, CCL2, and IL-6 were measured in the culture medium using standard dual-antibody solid phase immunoassay (ELISA) kits purchased from Peprotech. Cytokine/chemokine concentrations were normalized to total cellular protein levels, which were determined using the bicinchoninic acid protein assay as previously described[50].
Expression of inflammatory signaling molecules
Induction of inflammatory signaling was determined by measuring phosphorylation of inhibitory kappa B alpha (IκBα), and p38 mitogen-activated protein kinase (p38 MAPK) in cytoplasmic fractions; and phosphorylation of nuclear factor (NF)-κB p65 in nuclear fractions. More specifically, after experimental treatments, cells in 100 mm dishes were washed twice with PBS and nuclear and cytoplasmic protein extracts prepared as previously described[51], with the exception of including 1 mmol/L sodium orthovanadate (Sigma, #450243) in the lysis buffers. Thirty micrograms of total protein were loaded on 7.5% polyacrylamide gels (BioRad TGX FastCast Acrylamide kit, #161-0171), electrophoresed and then transferred to PVDF membrane. Membranes were then incubated at 4 °C for 15-18 h with rocking. Primary antibodies included anti-phospho-IκBα (1:100, Cell Signaling #2859S), anti-IκBα (1:1000, Cell Signaling #4812S), anti-phospho-p38 (1:500, Cell Signaling #9215S), anti-p38 (1:1000, Cell Signaling #9212S), anti-phospho-NF-κB p65 (1:1000, Cell Signaling #3033S), anti-NF-κB p65 (1:1000, Cell Signaling #4764S), and anti-β-tubulin (1:1000, Cell Signaling #2146S). Anti-rabbit IgG, AP-linked (1:1000, Cell Signaling #7054S) was used as the secondary antibody and restore western blot stripping buffer was used to remove antibodies and allow for re-labelling of membranes. The blots were scanned and analyzed as described above in the section on microglia marker expression.
Statistical analyses
Prism version 7.0 software (GraphPad Inc., San Diego, CA) was used for figure presentation and statistical analysis. Analyses included one-way analysis of variance (ANOVA), followed by Tukey’s multiple comparisons test. In those instances where data did not exhibit homogeneity of variance (P < 0.05 for Bartlett’s test), data were log-transformed prior to analysis. Data are presented as mean ± SEM (n = 4-7) and P < 0.05 indicated statistically significant differences between groups.
Results
Expression of microglial marker
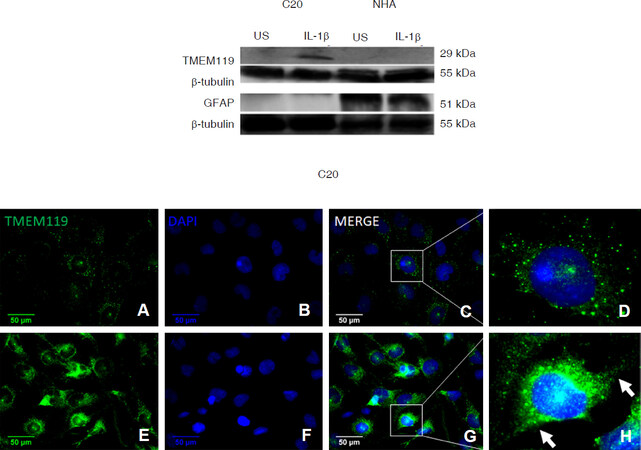
TMEM119 was robustly expressed in IL-1β-treated C20 cells as indicated by a strong band at ≈ 29 kDa [Figure 1 Top]. Whereas, TMEM119 was not detected in either unstimulated C20 cells or in NHA by western blot analysis. As expected, the astrocyte marker, GFAP was expressed in NHA, but was not detected in C20 cells.
Figure 1. C20 human microglial cells express the microglial marker transmembrane protein 119 (TMEM119). C20 cells were exposed to media alone (unstimulated; US) or media containing interleukin-1β (IL-1β) (20 ng/mL) for 24 h. Top panel: western blot analysis was used to measure levels of TMEM119 and glial fibrillary acidic protein (GFAP) in whole cell lysates and β-tubulin was assessed as a loading control. Whole cell lysates from unstimulated and IL-1β (3 ng/mL)-stimulated normal human astrocytes (NHA) were used for comparison. The blots presented are representative of independent experiments (n = 3 for C20; and n = 2 for NHA). Bottom panel: fluorescent immunocytochemistry was used to further assess TMEM119 expression (green) in US (A-D) and IL-1β-stimulated (E-H) C20 cells; and nuclei were labeled with 4ʹ,6-diamidino-2-phenylindole (DAPI, blue). Images are shown at 400 × magnification. The arrows in box H highlight the cytoplasmic extensions
Using fluorescent immunocytochemistry we confirmed our western blot findings that IL-1β-treated C20 cells express TMEM119 [Figure 1 Bottom]. This immunocytochemistry approach revealed that TMEM119 is also expressed in unstimulated C20 cells. In both unstimulated and IL-1β-stimulated cells, TMEM119 was detected predominantly in the cytoplasmic and/or cell membrane regions [Figure 1 Bottom]. Consistent with the western blot findings, TMEM119 expression was more pronounced in the IL-1β-treated C20 cells compared to unstimulated cells.
Cytokine/chemokine expression
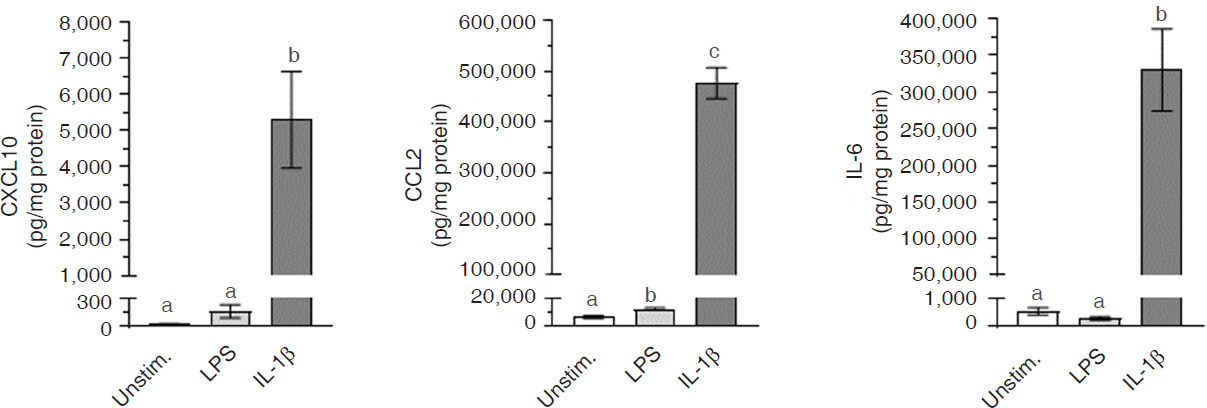
C20 cells constitutively expressed only minimal amounts of CXCL10, CCL2, and IL-6 [Figure 2]. However, stimulation with IL-1β significantly (P < 0.0001) induced expression of CXCL10, CCL2, and IL-6. In contrast to IL-1β, LPS induced only a minimal, yet significant (P < 0.01), increase in CCL2 expression, and did not significantly (P > 0.05) affect CXCL10 or IL-6 expression.
Figure 2. Cytokine/chemokine expression in C20 human microglial cells. C20 cells were exposed to media alone (unstimulated; Unstim.) or media containing either lipopolysaccharide (LPS) (1 µg/mL) or interleukin-1β (IL-1β) (20 ng/mL) for 24 h. CXCL10, CCL2, and IL-6 levels in the culture medium were determined by ELISA and normalized to total cellular protein (as determined by the bicinchoninic acid method). Data represent mean ± SEM (n = 4-7). Bars with different letters are significantly different (P < 0.01) as determined by one-way ANOVA and Tukey’s pairwise comparisons
Expression of inflammatory signaling molecules
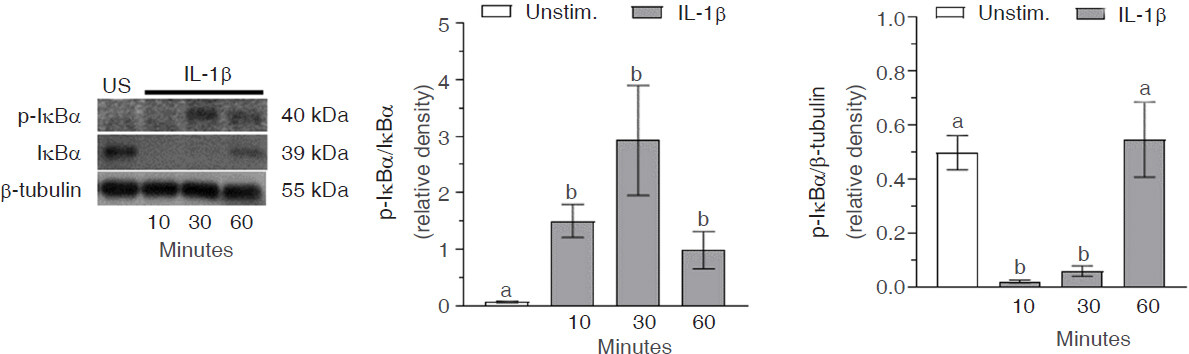
Levels of phosphorylated IκBα in the cytoplasm were significantly (P < 0.0001) increased after 10 min IL-1β exposure, remained elevated out to 60 min, but were beginning to drop toward baseline levels [Figure 3]. IκBα was constitutively expressed, with levels significantly (P < 0.0001) reduced 10 min after IL-1β treatment. IκBα expression remained significantly (P < 0.001) reduced 30 min after stimulation, but increased back to baseline levels by 60 min.
Figure 3. Interleukin-1β (IL-1β)-induced inhibitory kappa B alpha (IκBα) activation in C20 human microglial cells. C20 cells were exposed to media alone (unstimulated; US) or media containing IL-1β (20 ng/mL) for 10-60 min. Western blot analysis was used to measure levels of p-IκBα, IκBα, and β-tubulin in cytoplasmic protein extracts. The blots presented are representative of independent experiments (n = 4-5) and the data represent mean ± SEM. Bars with different letters are significantly different (P < 0.05) as determined by one-way ANOVA and Tukey’s pairwise comparisons
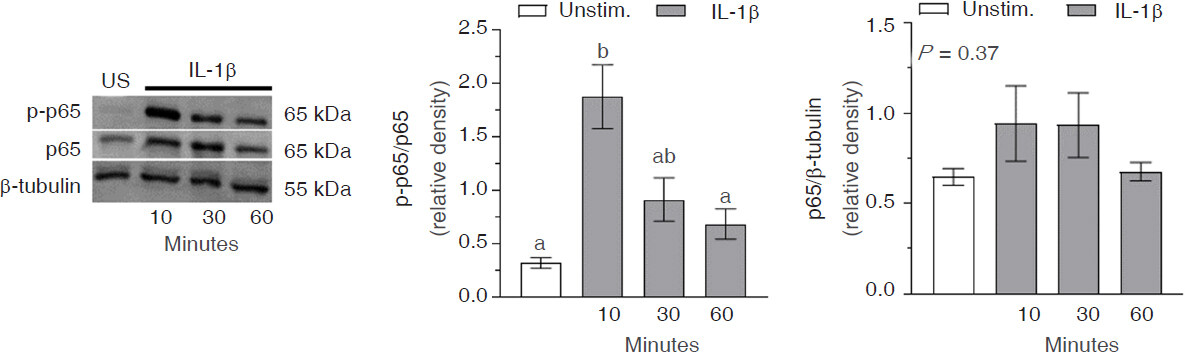
The expression of phospho-p65 in the nucleus rapidly increased within 10 min in response to IL-1β treatment (P < 0.0001), before declining to baseline levels by 30 min [Figure 4]. Constitutive expression of p65 in the nucleus of C20 cells was evident and levels remained unchanged (P = 0.37) throughout the 60 min exposure to IL-1β.
Figure 4. Interleukin-1β (IL-1β)-induced nuclear factor (NF)-κB p65 activation in C20 human microglial cells. C20 cells were exposed to media alone (unstimulated; US) or media containing IL-1β (20 ng/mL) for 10-60 min. Western blot analysis was used to measure levels of p-NF-κB p65, NF-κB p65, and β-tubulin in nuclear protein extracts. The blots presented are representative of independent experiments (n = 4-5) and the data represent mean ± SEM. Bars with different letters are significantly different (P < 0.05) as determined by one-way ANOVA and Tukey’s pairwise comparisons
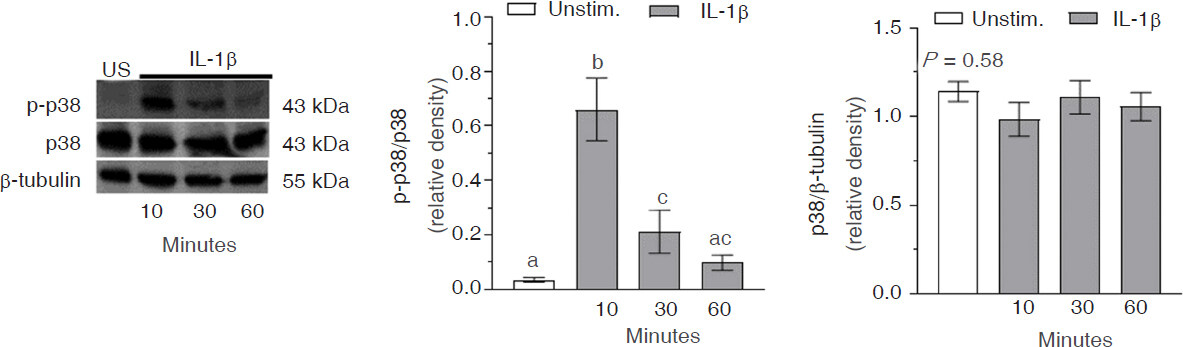
Phosphorylation of p38 in the cytoplasm occurred rapidly following treatment with IL-1β as indicated by a significant (P < 0.0001) increase in phospho-p38/p38 after 10 min [Figure 5]. The levels of phospho-p38 then rapidly declined by 30 min (P < 0.05), and returned to baseline by 60 min (P = 0.19). Constitutive expression of p38 in the cytoplasm of C20 cells was robust and the expression levels remained constant (P = 0.58) throughout the 60 min exposure to IL-1β.
Figure 5. Interleukin-1β (IL-1β)-induced p38 MAPK activation in C20 human microglial cells. C20 cells were exposed to media alone (unstimulated; US) or media containing IL-1β (20 ng/mL) for 10-60 min. Western blot analysis was used to measure levels of p-p38 mitogen-activated protein kinase (p38 MAPK), p38 MAPK, and β-tubulin in cytoplasmic protein extracts. The blots presented are representative of independent experiments (n = 5) and the data represent mean ± SEM. Bars with different letters are significantly different (P < 0.05) as determined by one-way ANOVA and Tukey’s pairwise comparisons
Discussion
Microglia are a key component of the innate immune system with critical roles in response to injury and infection[1]. Furthermore, microglia are instrumental in neurodevelopment and physiological functions necessary for maintaining CNS homeostasis[52]. Microglia have been implicated in a multitude of CNS disorders, and modulation of microglia function has emerged as a potential therapeutic strategy[8]. Altogether, there is substantial interest in further understanding microglia function. Much of what is currently known about microglia, stems from in vitro studies using rodent microglia, particularly immortalized cell lines. However, there is increasing interest in the use of immortalized human microglial cell lines to advance our understanding of human microglia function and discovery of pharmacological agents that modulate microglia[37,38,53,54].
Among the very few immortalized human microglial cell lines available for use is the C20 human microglial cell line recently developed by Garcia-Mesa et al.[37]. These investigators utilized RNA sequencing to confirm the microglial phenotype of C20 cells and they demonstrated that these cells maintain migratory capacity and phagocytic activity characteristic of microglia[37]. Furthermore, C20 cells express numerous microglial surface markers, including cluster of differentiation (CD)11b, CD68, TGFβ receptor (TGFβR), and the P2 purinergic receptor, P2RY12, as indicated by immunofluorescence and flow cytometry[37]. While CD11b+ macrophages are also present in peripheral tissues[55], TGFβR and P2RY12 have been suggested to be microglial specific[56,57]. TMEM119 has also recently emerged as a microglia marker capable of discriminating between resident microglia and peripheral macrophages[46,47], yet the functional importance of this protein remains to be elucidated. We have demonstrated for the first time that C20 cells express TMEM119. Interestingly, TMEM119 protein expression was greatest in C20 cells that were stimulated with IL-1β. While we did detect very small amounts of TMEM119 in unstimulated C20 cells during preliminary experiments, a substantial amount of total protein had to be loaded into the gels in order to achieve these results. Whereas, when 30 µg of protein from unstimulated C20 cells were electrophoresed, we did not detect TMEM119. Further investigation is warranted to fully appreciate the expression profile of TMEM119 at the mRNA and protein levels; and the modulatory influence of proinflammatory mediators. Additional insights into the expression profile and functional role of TMEM119 in microglia are therefore needed and C20 cells may prove to be a useful tool in this line of investigation.
Previously, during the initial characterization of this cell line, C20 cells were found to secrete proinflammatory cytokines following stimulation with TNFα[37]. We have added to these findings and advanced our understanding of inflammatory signaling in C20 cells. More specifically, we provide the first evidence that IL-1β potently induces expression of CXCL10, CCL2, and IL-6 in C20 cells. We have also demonstrated that LPS differentially affects expression of these inflammatory mediators as evidenced by stimulation of CCL2 expression, but not CXCL10 or IL-6. Overall, it is clear that IL-1β is a much more effective inducer of cytokine/chemokine expression in C20 cells compared to LPS. In contrast, mouse microglia (including the BV-2 cells) are very responsive to LPS, but not to IL-1β due to the absence of IL-1R expression[36].
IL-1β levels are elevated in a range of CNS disorders and this proinflammatory cytokine has been implicated as a key mediator of neuropathology[40]. Thus, it is fundamentally important to fully understand IL-1β-induced inflammatory signaling in human microglia. Furthermore, advancing our understanding of these signaling events in C20 human microglia is critical for developing this research tool. Secretion of CXCL10, CCL2 and IL-6 by C20 microglia in response to IL-1β exposure is a functionally relevant endpoint measure given both the important neurophysiological and neuropathological roles of these cytokines/chemokines. For instance, CXCL10 is initially neuroprotective in viral infections[58], but can also contribute to neuropathology as evidenced in human immunodeficiency virus (HIV)-induced dementia[59,60]. CXCL10 also plays a role in neuropathology associated with traumatic brain injury (TBI)[61], and emerging data suggest CXCL10 is involved in sickness behavior[62]. CCL2 functions as a chemotactic cytokine, activating and directing migration of numerous cell types[61]; and it is increasingly evident that increased levels of this chemokine in the brain contribute to neuropathology of HIV-dementia, AD, ischemia, epilepsy, and TBI[61]. The cytokine IL-6 acts in the hypothalamus as a regulator of metabolism[63] and has gained attention for its involvement in autism[64], major depression[65,66], and neurodegenerative diseases[67-69]. Therefore, C20 microglia are expected to be a useful tool in discovery of pharmacologic agents that may modulate microglial activation and subsequent release of proinflammatory factors.
We also provide the first evidence that IL-1β induces activation of key proinflammatory signaling molecules in C20 cells, including IκBα, NF-κB p65, and p38 MAPK. The importance of these signaling molecules in microglia activation is well established and these proteins are viable targets for pharmacologic modulation of inflammation[29,34,70-73]. Therefore, by demonstrating that these signaling pathways are functional in C20 cells, it is expected that these cells will be instrumental in the identification and characterization of novel pharmacologic agents intended to alter microglial function.
In conclusion, we have determined that IL-1β-activated C20 microglia express the microglia specific marker TMEM119. Additionally, we have provided the first evidence that IL-1β induces activation of IκBα, NF-κB p65, and p38 MAPK and subsequent secretion of CXCL10, CCL2 and IL-6 in C20 human microglia. These findings support the use of this human microglial cell line as a research tool to advance our understanding of microglia function and for the development of pharmacotherapies targeting a range of neuropathologies.
Declarations
AcknowledgementsWe greatly appreciate the technical assistance of Robert W. Allen, PhD and Jun Fu, PhD at the Human Identity Testing Laboratory in the School of Forensic Sciences at Oklahoma State University Center for Health Sciences.
Authors’ contributionsConcept, experimental design, literature review, statistical analysis, manuscript preparation: Davis RL
Performed experiments, data acquisition and analysis, and manuscript editing: Buck DJ
Performed experiments, experimental design, data acquisition and analysis, and manuscript editing: McCracken K
Performed experiments, data acquisition, and manuscript editing: Cox GW
Performed experiments, data acquisition and analysis, and manuscript editing: Das S
Availability of data and materialsThe raw data and materials are housed in the laboratory of Davis RL and available as appropriate.
Financial support and sponsorshipOklahoma Center for the Advancement of Science & Technology (HR14-007) (RLD); Oklahoma State University Center for Health Sciences, Intramural funds (RLD).
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2018.
REFERENCES
1. Wolf SA, Boddeke HW, Kettenmann H. Microglia in Physiology and Disease. Annu Rev Physiol 2017;79:619-43.
2. Nimmerjahn A, Kirchhoff F, Helmchen F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 2005;308:1314-8.
3. Davalos D, Grutzendler J, Yang G, Kim JV, Zuo Y, et al. ATP mediates rapid microglial response to local brain injury in vivo. Nat Neurosci 2005;8:752-8.
4. Hong S, Dissing-Olesen L, Stevens B. New insights on the role of microglia in synaptic pruning in health and disease. Curr Opin Neurobiol 2016;36:128-34.
5. Werneburg S, Feinberg PA, Johnson KM, Schafer DP. A microglia-cytokine axis to modulate synaptic connectivity and function. Curr Opin Neurobiol 2017;47:138-45.
6. Rogers JT, Morganti JM, Bachstetter AD, Hudson CE, Peters MM, et al. CX3CR1 deficiency leads to impairment of hippocampal cognitive function and synaptic plasticity. J Neurosci 2011;31:16241-50.
7. Schafer DP, Lehrman EK, Stevens B. The “quad-partite” synapse: microglia-synapse interactions in the developing and mature CNS. Glia 2013;61:24-36.
8. Salter MW, Stevens B. Microglia emerge as central players in brain disease. Nat Med 2017;23:1018-27.
9. Heneka MT, Kummer MP, Latz E. Innate immune activation in neurodegenerative disease. Nat Rev Immunol 2014;14:463-77.
10. Lee SC, Liu W, Dickson DW, Brosnan CF, Berman JW. Cytokine production by human fetal microglia and astrocytes. Differential induction by lipopolysaccharide and IL-1 beta. J Immunol 1993;150:2659-67.
11. Rustenhoven J, Park TI, Schweder P, Scotter J, Correia J, et al. Isolation of highly enriched primary human microglia for functional studies. Sci Rep 2016;6:19371.
12. Lokensgard JR, Hu S, van Fenema EM, Sheng WS, Peterson PK. Effect of thalidomide on chemokine production by human microglia. J Infect Dis 2000;182:983-7.
13. D’Aversa TG, Yu KO, Berman JW. Expression of chemokines by human fetal microglia after treatment with the human immunodeficiency virus type 1 protein Tat. J Neurovirol 2004;10:86-97.
14. Liu J, Zhao ML, Brosnan CF, Lee SC. Expression of type II nitric oxide synthase in primary human astrocytes and microglia: role of IL-1beta and IL-1 receptor antagonist. J Immunol 1996;157:3569-76.
15. Park J, Min JS, Kim B, Chae UB, Yun JW, et al. Mitochondrial ROS govern the LPS-induced pro-inflammatory response in microglia cells by regulating MAPK and NF-kappaB pathways. Neurosci Lett 2015;584:191-6.
16. Cianciulli A, Dragone T, Calvello R, Porro C, Trotta T, et al. IL-10 plays a pivotal role in anti-inflammatory effects of resveratrol in activated microglia cells. Int Immunopharmacol 2015;24:369-76.
17. Vinoth Kumar R, Oh TW, Park YK. Anti-inflammatory effects of Ginsenoside-Rh2 inhibits LPS-induced activation of microglia and overproduction of inflammatory mediators via modulation of TGF-beta1/Smad pathway. Neurochem Res 2016;41:951-7.
19. Pena-Altamira E, Prati F, Massenzio F, Virgili M, Contestabile A, et al. Changing paradigm to target microglia in neurodegenerative diseases: from anti-inflammatory strategy to active immunomodulation. Expert Opin Ther Targets 2016;20:627-40.
20. Jucaite A, Svenningsson P, Rinne JO, Cselenyi Z, Varnas K, et al. Effect of the myeloperoxidase inhibitor AZD3241 on microglia: a PET study in Parkinson’s disease. Brain 2015;138:2687-700.
21. Husain MI, Chaudhry IB, Husain N, Khoso AB, Rahman RR, et al. Minocycline as an adjunct for treatment-resistant depressive symptoms: a pilot randomised placebo-controlled trial. J Psychopharmacol 2017;31:1166-75.
22. Moller T, Bard F, Bhattacharya A, Biber K, Campbell B, et al. Critical data-based re-evaluation of minocycline as a putative specific microglia inhibitor. Glia 2016;64:1788-94.
23. Stansley B, Post J, Hensley K. A comparative review of cell culture systems for the study of microglial biology in Alzheimer’s disease. J Neuroinflammation 2012;9:115.
24. Horvath RJ, Nutile-McMenemy N, Alkaitis MS, Deleo JA. Differential migration, LPS-induced cytokine, chemokine, and NO expression in immortalized BV-2 and HAPI cell lines and primary microglial cultures. J Neurochem 2008;107:557-69.
25. Sarkar S, Malovic E, Sarda D, Lawana V, Rokad D, et al. Characterization and comparative analysis of a new mouse microglial cell model for studying neuroinflammatory mechanisms during neurotoxic insults. Neurotoxicology 2018;67:129-40.
26. Periyasamy P, Liao K, Kook YH, Niu F, Callen SE, et al. Cocaine-mediated downregulation of miR-124 activates microglia by targeting KLF4 and TLR4 signaling. Mol Neurobiol 2018;55:3196-210.
27. Lam D, Lively S, Schlichter LC. Responses of rat and mouse primary microglia to pro- and anti-inflammatory stimuli: molecular profiles, K(+) channels and migration. J Neuroinflammation 2017;14:166.
28. Yuan L, Liu S, Bai X, Gao Y, Liu G, et al. Oxytocin inhibits lipopolysaccharide-induced inflammation in microglial cells and attenuates microglial activation in lipopolysaccharide-treated mice. J Neuroinflammation 2016;13:77.
29. Wang H, Liu C, Han M, Cheng C, Zhang D. TRAM1 promotes microglia M1 polarization. J Mol Neurosci 2016;58:287-96.
30. Cao Q, Karthikeyan A, Dheen ST, Kaur C, Ling EA. Production of proinflammatory mediators in activated microglia is synergistically regulated by Notch-1, glycogen synthase kinase (GSK-3beta) and NF-kappaB/p65 signalling. PLoS One 2017;12:e0186764.
31. Melief J, Sneeboer MA, Litjens M, Ormel PR, Palmen SJ, et al. Characterizing primary human microglia: a comparative study with myeloid subsets and culture models. Glia 2016;64:1857-68.
32. Si Q, Zhao ML, Morgan AC, Brosnan CF, Lee SC. 15-deoxy-Delta12,14-prostaglandin J2 inhibits IFN-inducible protein 10/CXC chemokine ligand 10 expression in human microglia: mechanisms and implications. J Immunol 2004;173:3504-13.
33. Peferoen LA, Vogel DY, Ummenthum K, Breur M, Heijnen PD, et al. Activation status of human microglia is dependent on lesion formation stage and remyelination in multiple sclerosis. J Neuropathol Exp Neurol 2015;74:48-63.
34. Olajide OA, Aderogba MA, Fiebich BL. Mechanisms of anti-inflammatory property of Anacardium occidentale stem bark: inhibition of NF-kappaB and MAPK signalling in the microglia. J Ethnopharmacol 2013;145:42-9.
35. McManus CM, Brosnan CF, Berman JW. Cytokine induction of MIP-1 alpha and MIP-1 beta in human fetal microglia. J Immunol 1998;160:1449-55.
36. Pinteaux E, Parker LC, Rothwell NJ, Luheshi GN. Expression of interleukin-1 receptors and their role in interleukin-1 actions in murine microglial cells. J Neurochem 2002;83:754-63.
37. Garcia-Mesa Y, Jay TR, Checkley MA, Luttge B, Dobrowolski C, et al. Immortalization of primary microglia: a new platform to study HIV regulation in the central nervous system. J Neurovirol 2017;23:47-66.
38. Nagai A, Nakagawa E, Hatori K, Choi HB, McLarnon JG, et al. Generation and characterization of immortalized human microglial cell lines: expression of cytokines and chemokines. Neurobiol Dis 2001;8:1057-68.
39. Janabi N, Peudenier S, Heron B, Ng KH, Tardieu M. Establishment of human microglial cell lines after transfection of primary cultures of embryonic microglial cells with the SV40 large T antigen. Neurosci Lett 1995;195:105-8.
40. Mendiola AS, Cardona AE. The IL-1beta phenomena in neuroinflammatory diseases. J Neural Transm (Vienna) 2018;125:781-95.
41. Facci L, Barbierato M, Zusso M, Skaper SD, Giusti P. Serum amyloid A primes microglia for ATP-dependent interleukin-1beta release. J Neuroinflammation 2018;15:164.
42. Sun M, Brady RD, Wright DK, Kim HA, Zhang SR, et al. Treatment with an interleukin-1 receptor antagonist mitigates neuroinflammation and brain damage after polytrauma. Brain Behav Immun 2017;66:359-71.
43. Spencer SJ, D’Angelo H, Soch A, Watkins LR, Maier SF, et al. High-fat diet and aging interact to produce neuroinflammation and impair hippocampal- and amygdalar-dependent memory. Neurobiol Aging 2017;58:88-101.
44. Figueroa-Hall LK, Anderson MB, Das S, Stevens CW, Davis RL. LPS-induced TLR4 neuroinflammatory sugnaling in CHME-5 microglial cells. Neuroimmunol Neuroinflammation 2017;4:219-31.
45. Davis RL, Das S, Buck DJ, Stevens CW. Beta-funaltrexamine inhibits chemokine (CXCL10) expression in normal human astrocytes. Neurochem Int 2013;62:478-85.
46. Satoh J, Kino Y, Asahina N, Takitani M, Miyoshi J, et al. TMEM119 marks a subset of microglia in the human brain. Neuropathology 2016;36:39-49.
47. Bennett ML, Bennett FC, Liddelow SA, Ajami B, Zamanian JL, et al. New tools for studying microglia in the mouse and human CNS. Proc Natl Acad Sci U S A 2016;113:E1738-46.
48. Attaai A, Neidert N, von Ehr A, Potru PS, Zoller T, et al. Postnatal maturation of microglia is associated with alternative activation and activated TGFbeta signaling. Glia 2018; doi: 10.1002/glia.23332.
49. Zrzavy T, Hoftberger R, Berger T, Rauschka H, Butovsky O, et al. Pro-inflammatory activation of microglia in the brain of patients with sepsis. Neuropathol Appl Neurobiol 2018; doi: 10.1111/nan.12502.
50. Davis RL, Dertien J, Syapin PJ. Ethanol-induced modulation of inducible nitric-oxide synthase activity in human A172 astrocytoma cells. Alcohol Clin Exp Res 2002;26:1404-11.
51. Sanchez AC, Davis RL, Syapin PJ. Identification of cis-regulatory regions necessary for robust Nos2 promoter activity in glial cells: indirect role for NF-kappaB. J Neurochem 2003;86:1379-90.
52. Sominsky L, De Luca S, Spencer SJ. Microglia: key players in neurodevelopment and neuronal plasticity. Int J Biochem Cell Biol 2018;94:56-60.
53. Jadhav VS, Krause KH, Singh SK. HIV-1 Tat C modulates NOX2 and NOX4 expressions through miR-17 in a human microglial cell line. J Neurochem 2014;131:803-15.
54. Branca JJ, Morucci G, Malentacchi F, Gelmini S, Ruggiero M, et al. Effects of oxaliplatin and oleic acid Gc-protein-derived macrophage-activating factor on murine and human microglia. J Neurosci Res 2015;93:1364-77.
55. Bain CC, Bravo-Blas A, Scott CL, Perdiguero EG, Geissmann F, et al. Constant replenishment from circulating monocytes maintains the macrophage pool in the intestine of adult mice. Nat Immunol 2014;15:929-37.
56. Butovsky O, Jedrychowski MP, Moore CS, Cialic R, Lanser AJ, et al. Identification of a unique TGF-beta-dependent molecular and functional signature in microglia. Nat Neurosci 2014;17:131-43.
57. Zhu C, Kros JM, van der Weiden M, Zheng P, Cheng C, et al. Expression site of P2RY12 in residential microglial cells in astrocytomas correlates with M1 and M2 marker expression and tumor grade. Acta Neuropathol Commun 2017;5:4.
58. Lokensgard JR, Mutnal MB, Prasad S, Sheng W, Hu S. Glial cell activation, recruitment, and survival of B-lineage cells following MCMV brain infection. J Neuroinflammation 2016;13:114.
59. Sanfilippo C, Pinzone MR, Cambria D, Longo A, Palumbo M, et al. OAS gene family expression is associated with HIV-related neurocognitive disorders. Mol Neurobiol 2018;55:1905-14.
60. Mehla R, Bivalkar-Mehla S, Nagarkatti M, Chauhan A. Programming of neurotoxic cofactor CXCL-10 in HIV-1-associated dementia: abrogation of CXCL-10-induced neuro-glial toxicity in vitro by PKC activator. J Neuroinflammation 2012;9:239.
61. Gyoneva S, Ransohoff RM. Inflammatory reaction after traumatic brain injury: therapeutic potential of targeting cell-cell communication by chemokines. Trends Pharmacol Sci 2015;36:471-80.
62. Davis RL, Stevens CW, Thomas Curtis J. The opioid antagonist, beta-funaltrexamine, inhibits lipopolysaccharide-induced neuroinflammation and reduces sickness behavior in mice. Physiol Behav 2017;173:52-60.
63. Timper K, Denson JL, Steculorum SM, Heilinger C, Engstrom-Ruud L, et al. IL-6 Improves energy and glucose homeostasis in obesity via enhanced central IL-6 trans-signaling. Cell Rep 2017;19:267-80.
65. Furtado M, Katzman MA. Examining the role of neuroinflammation in major depression. Psychiatry Res 2015;229:27-36.
66. Engler H, Brendt P, Wischermann J, Wegner A, Rohling R, et al. Selective increase of cerebrospinal fluid IL-6 during experimental systemic inflammation in humans: association with depressive symptoms. Mol Psychiatry 2017;22:1448-54.
67. Lindqvist D, Hall S, Surova Y, Nielsen HM, Janelidze S, et al. Cerebrospinal fluid inflammatory markers in Parkinson’s disease--associations with depression, fatigue, and cognitive impairment. Brain Behav Immun 2013;33:183-9.
68. Chen SY, Chen TF, Lai LC, Chen JH, Sun Y, et al. Sequence variants of interleukin 6 (IL-6) are significantly associated with a decreased risk of late-onset Alzheimer’s disease. J Neuroinflammation 2012;9:21.
69. Spooren A, Kolmus K, Laureys G, Clinckers R, De Keyser J, et al. Interleukin-6, a mental cytokine. Brain Res Rev 2011;67:157-83.
70. Yuan T, Li Z, Li X, Yu G, Wang N, et al. Lidocaine attenuates lipopolysaccharide-induced inflammatory responses in microglia. J Surg Res 2014;192:150-62.
71. Weng L, Zhang H, Li X, Zhan H, Chen F, et al. Ampelopsin attenuates lipopolysaccharide-induced inflammatory response through the inhibition of the NF-kappaB and JAK2/STAT3 signaling pathways in microglia. Int Immunopharmacol 2017;44:1-8.
72. Choi MJ, Lee EJ, Park JS, Kim SN, Park EM, et al. Anti-inflammatory mechanism of galangin in lipopolysaccharide-stimulated microglia: critical role of PPAR-gamma signaling pathway. Biochem Pharmacol 2017;144:120-31.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Davis RL, Buck DJ, McCracken K, Cox GW, Das S. Interleukin-1β-induced inflammatory signaling in C20 human microglial cells. Neurosciences 2018;5:50. http://dx.doi.org/10.20517/2347-8659.2018.60
AMA Style
Davis RL, Buck DJ, McCracken K, Cox GW, Das S. Interleukin-1β-induced inflammatory signaling in C20 human microglial cells. Neuroimmunology and Neuroinflammation. 2018; 5: 50. http://dx.doi.org/10.20517/2347-8659.2018.60
Chicago/Turabian Style
Davis, Randall L., Daniel J. Buck, Kelly McCracken, Gary W. Cox, Subhas Das. 2018. "Interleukin-1β-induced inflammatory signaling in C20 human microglial cells" Neuroimmunology and Neuroinflammation. 5: 50. http://dx.doi.org/10.20517/2347-8659.2018.60
ACS Style
Davis, RL.; Buck DJ.; McCracken K.; Cox GW.; Das S. Interleukin-1β-induced inflammatory signaling in C20 human microglial cells. Neurosciences. 2018, 5, 50. http://dx.doi.org/10.20517/2347-8659.2018.60
About This Article
Copyright
Data & Comments
Data
 Cite This Article 30 clicks
Cite This Article 30 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.