Management strategies in acute traumatic spinal cord injury: a narrative review
Abstract
Worldwide, spinal cord injury (SCI) affects around 500,000 people each year and results in significant morbidity. The primary insult to the spinal cord occurs at the time of the initial injury, which may result from a contusion, laceration or more rarely a transection. Secondary damage in SCI is more insidious and subacute; it is the result of a combination of an inflammatory response, vascular changes and ionic dysregulation. Early clinical intervention is vital after the acute, primary insult to ensure the best possible outcomes for these patients. Current evidence on the demographics and mechanisms, underlying basic science and management strategies of spinal cord injuries are outlined.
Keywords
Introduction
Worldwide, spinal cord injury (SCI) is a significant pathology that affects around 500,000 people each year. The injury itself results in significant morbidity.
These injuries are typically of a traumatic aetiology and carry significant impairment to function and quality of life. 40.4% are involved in road traffic accidents, 27.9% in falls, 8% are sports injuries, 15% related to violence and 8.5% are due to tumours or other causes. The burden of these injuries to both the patient and society as a whole is significant. The financial cost for each individual with such an injury is US$3 million with an estimated overall annual cost of $10 billion per year[1].
Primary SCI occurs at the time of initial insult to the cord, which may result from a contusion, laceration or more rarely a transection. Maximal neurological deficit is observed immediately after a SCI and this results from the loss of effective axonal transmission, which is hampered by neuronal damage, damage to endothelial cells, ongoing haemorrhage and shifts in ionic concentrations.
Secondary damage in SCI is more insidious and subacute[1]. It results from the combination of an inflammatory response, vascular changes and ionic dysregulation. As such, timely intervention after the acute primary insult is vital to ensure the best possible outcomes for these patients.
The often-permanent functional impairment in the injured spinal cord is due to poor healing potential. This contrasts to the repair and functional recovery of other tissues such as skin and muscles. The reason for this is not entirely understood; however, as described below, inflammation likely plays a key role[2].
Obviously, this adds additional necessity for timely intervention and appropriate treatment, not least for the patients themselves, but for society at large.
Current evidence on the demographics and mechanisms of SCIs, the basic science of SCIs, and management strategies are outlined.
The authors aim to perform a narrative review of SCI. Basic science and pathophysiology, mechanisms, management strategies and current best evidence will all be presented to offer a rounded and thorough review of SCIs and their management, for both scientific and clinical reference.
Basic science of spinal cord injury
The spinal cord consists of many multitudes of neurons, which are the component active cell in the central and peripheral nervous systems. Neurons, while there is a certain variety in morphology, contain the following components: cell body, dendrites, axon and axon terminals. The cell body contains the nucleus and contains neuronal proteins and membranes. Axons, coupled with axon terminals, function to relay electrical impulses known as action potentials to stimulate responses in the central nervous system. Axons are layered with myelin sheaths, which enable swifter transmission of action potentials. Dendrites extend out from the cell body, which act to receive impulses from other neuron axons. These are extremely long, particularly in the central nervous system, and have many complex interactions with other neurons[3].
Neuron progentior cells are progenitor cells within the CNS that result in the glial and neuronal cell types that populate the CNS. NPCs have no role in generating the non-neural cells that are also present in the CNS, such as immune system cells. NPCs are present in the developing embryo’s CNS but are also found in the neonatal and mature adult brain[4].
Astrocytes are among the number of glial cells. They are ectodermal neural cells that maintain homeostasis and help defend the central nervous system. They are heterogeneous in form and function and have adaptive plasticity that defines the functional maintenance of the CNS with growth and age. They transport major ions and protons, remove and catabolize neurotransmitters, and release neurotransmitter precursors and scavengers of reactive oxygen species[5].
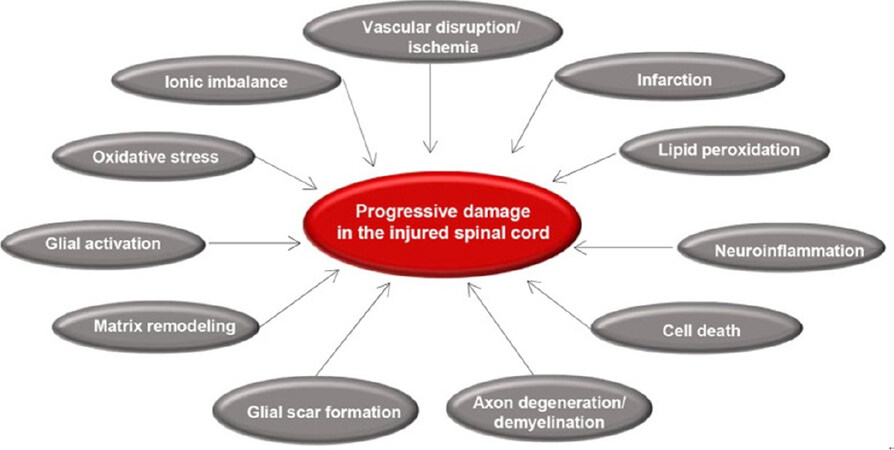
A SCI can be divided into (1) a primary injury which has occurred as a direct result of the initial insult; and (2) an ensuing secondary injury which is more insidious and subacute[1]. It results from the combination of an inflammatory response, vascular changes and ionic dysregulation (all explored in Figure 1). Primary SCI occurs from a series of direct insults. Transection, whereby the spinal cord is transected by a blunt or sharp force, can be complete or partial. Any ascending or descending neuronal tissue will be damaged by such an injury. Contusion of the spinal cord occurs after a transient physical impact. This can result in compression or impact related damage to the cord. The fundamental factor in contusion is that the impact is transient and brief. Compression is a contusion-type injury following by prolonged force application over time[6].
Figure 1. Primary and secondary mechanisms of spinal cord injury. Adapted from Alizadeh et al.[41]
All primary SCIs will result in secondary damage
Inflammatory response
The spinal cord is not exposed to inflammatory processes in a healthy individual and as a result, when inflammation does occur, it can have devastating consequences. The spinal cord is separated from ongoing inflammatory cells within the body by endothelial cells, which form a physical barrier[7]. The inflammatory response is initiated by damage to these endothelial cells, which leads to an increase in permeability and intracellular oedema, both of which are key factors in recruiting pro-inflammatory cells, leading to ongoing secondary injury[8].
The initiation of this inflammatory response is an almost immediate consequence of SCI. The inflammatory response is mediated by pro-inflammatory cytokines including IL1B, IL6 and TNF-alpha, which are released from damaged endothelial cells[9]. TNF-alpha induces damage in acute inflammation through stimulating apoptosis and necrosis. It is produced by activated macrophages. IL1B increases the levels of TNF-α. This results in reduced neuronal survival, exacerbating lesion size and astrogliosis, and dampening axonal plasticity[10]. IL6 promotes the infiltration and activation of mononuclear leucocytes while suppressing neutrophil infiltration[11].
These inflammatory cytokines are very much involved in the acute phase of the injury. It has been found in histochemical analysis of human patients with SCIs, IL1B, IL6 and TNF-alpha were detected in neurons within thirty minutes of an acute SCI. These levels declined within two days after the injury[12].
Other studies have shown that these cells increase in the first four days following the injury[13]. This acute inflammatory phase is a transient process lasting approximately ten days following SCI, and end at 3 weeks following injury with inflammatory cells becoming absent from CSF then to reflect the end of ongoing secondary damage[9].
Cellular role
Remyelination plays an important role in the recovery of axons after SCI. While pro-inflammatory cytokines mediate inflammation and cell damage in SCI, there is a parallel action of proliferation-orientated cells.
Microglia are a key cellular component of the scar that develops after SCI to protect neural tissue. They are dynamic and proliferate extensively during the first two weeks, accumulating around the lesion. There, microglia move to the interface between infiltrating leukocytes and astrocytes, where they proliferate and form a scar[14].
Oligodendrocyte precursor cells (OPCs) regulate the inflammatory reaction after SCI. After injury, OPCs migrate to the injury site and rapidly proliferate. From the day of injury to day 7, the number of OPCs persistently increases.
Activated astrocytes influence proliferation, differentiation, and maturation of inflammatory reactions after injury. Astrocytes are also involved in synaptogenesis and control the immune response. It is these factors that play a fundamental role in remyelination after acute SCI[15]. After trauma, astrocytes surrounding the lesion become reactive and become hypertrophic. They migrate centripetally to the epicentre of the lesion and help with the tissue repair process. These reactive astrocytes do eventually become scar-forming astrocytes and form a glial scar. Within and around the glial scar, cells deposit extracellular matrix proteins that affect axon growth[16]. This can result in axonal growth inhibitors and then prevent axonal regeneration. Glial scar formation is one of the main causes of the limited regenerative capability of the CNS[17].
Macrophage activation also plays an important cellular role in regulating neuronal damage in the injured spinal cord. Macrophages have the ability to promote the repair of injured tissue by regulating transitions through the different phases of the healing response. In the injured spinal cord, pro-inflammatory macrophages potentiate a prolonged inflammatory phase and remodelling is not properly initiated[18].
Vascular changes
Mechanical damage to the spinal cord results in immediate vasospasm of superficial vessels and intraparenchymal haemorrhage. This damage initially occurs in the highly vascularised, yet most vulnerable grey matter[10]. This leads to immediate mechanical damage to the grey matter microvasculature, which further impairs the microcirculation to the cord and impedes perfusion[19]. The impaired blood flow of the damaged spinal cord may then beecome further damaged by systemic responses to the injury such as hypotension, bradycardia and a decreased CO2, leading to further ischemic damage[13].
Free radical damage
Cells under stress, in pro-inflammatory states such as the acutely injured spinal cord, generate large quantities of free radicals. These reactive species lead to ionic dysregulation when generated in excess. They can overload and block normal cellular signalling pathways. Impaired electron pumps such as Na+/K+/ATPase causes increased intracellular calcium. This leads to apoptosis, as well as mitochondrial dysfunction, contributing to ongoing spinal cord damage[20,21]. Redox potentials within the cells then plummet and result in oxidative damage. Such oxidative damage can continue for up to five days following the initial injury, contributing to the pathogenesis of secondary injury. Proteins and nucleic acids are damaged by the free radicals from red-ox reactions, leading to further ongoing damage to the spinal cord[10].
Management strategies in spinal cord injury
Management strategies for acute SCIs are typically focused on negating any secondary insult, mediated by the vascular, inflammatory and free radical changes after the primary injury. A thorough grounding in the mechanisms described above is therefore essential for guiding appropriate management.
Cardiovascular support
Cardiovascular support for acute SCIs is essential in maintaining spinal cord perfusion after a traumatic injury. As described, physical damage to the cord results in immediate vasospasm of the microvasculature of the cord. Maintaining an adequate mean arterial pressure optimises cord perfusion. In particular, patients with complete high cervical SCIs are likely to develop spinal shock with loss of sympathetic drive. This results in hypotension due to the loss of peripheral vascular tone and concomitant bradycardia[22]. These patients are more likely to require vasopressor support to maintain their mean arterial pressure at the required levels, compared to incomplete injuries and those with thoracic or lumbar levels of injury (P = 0.001)[23].
An observational study of 91 patients demonstrated that spinal cord perfusion pressure is an independent predictor of neurologic recovery in acute SCI [odds ratio (OR) = 1.039, P = 0.002][24]. These study findings support the need for vasopressor support in acute SCI.
High levels of evidence are not available but cohort studies have demonstrated improvement in neurologic outcomes in patients with high average mean arterial pressure values. A mean arterial pressure of 85-90 mmHg is appropriate[25-27]. The duration of vasopressor support however, does not appear to be absolutely supported in the literature. A systematic review of vasopressor support did mention that a duration of five to seven days should be considered. The choice of vasopressor though does appear to be important in some cohorts. A retrospective cohort analysis of 34 patients in California showed that in a subgroup of patients over 55 years of age, dopamine produced statistically significant increases in the complication rates when compared with phenylephrine [83% vs. 50% for dopamine and phenylephrine, respectively; OR with dopamine 5.0 (95%CI: 0.99-25.34), P = 0.044][28]. This subgroup of 34 patients also demonstrated a median improvement of one ASIA grade from admission to discharge, with no difference between vasopressor agents.
Steroid administration
Steroids have traditionally been given in acute SCI. The hypothesis is that steroids reduce inflammation and prevent secondary cord injury. However, a recent meta-analysis has debunked the evidence for their routine use. A Cochrane review of three randomised controlled trials has shown no difference in neurology between treatment and placebo groups at six and twelve months post-injury[29]. Nevertheless, despite the lack of evidence for routine administration of steroids in acute SCI, they appear to be routinely administered in many institutions still. The rationale for this is varied, but the fear of medico-legal consequences is one such reason. In the same study, surgeons also felt that there was little risk associated with the routine administration of these treatments regardless, despite minimal demonstrated clinical benefit[30].
Surgical treatment
Evidence suggests that decompression within 24 h of injury carries the greatest potential improvement in neurologic function for patients with incomplete SCI after trauma.
The timing of surgical decompression is a factor that plays a role in neurologic recovery. There has been some debate and certainly, this is a factor in the trauma patient with SCI and multiple concomitant injuries, particularly chest injuries. Indeed, as discussed later, there is some evidence to suggest conservative management will result in neurologic recovery also.
In a recent meta-analysis of nine studies, patients with traumatic SCIs who were decompressed within 24 hours had a significant neurologic improvement rate (OR = 1.66, 95%CI: 1.19-2.31, P < 0.01), a shorter length of hospital stay by almost five days (P = 0.04) as well as fewer post-operative complications (OR = 0.61, 95%CI: 0.40-0.91, P = 0.02)[31]. Surgery within 24 h for acute traumatic SCI is thus superior to delaying surgery for neurologic outcomes.
In another meta-analysis performed by Ter-Wengel et al.[32], 422 patients with complete cervical traumatic SCI showed that improvement was more likely after early surgery [respectively, 22.6%, 95%CI: 16.6%-28.7% and 10.4%, 95%CI: 5.6%-15.8%; OR = 2.6 (95%CI: 1.4-5.1)]. The same meta-analysis showed that in 636 patients with incomplete cervical traumatic SCI, there were no differences between early or late surgery. The authors thus suggest a shift in the treatment of patients with complete cervical traumatic SCI. The authors’ previous understanding of the literature was that there was equivocal evidence for recovery in complete traumatic cervical SCI. The findings from this study changed that previously held position, in favour of early surgical decompression. In incomplete cervical traumatic SCI, neurological outcome is similar between early and late surgery[32].
In traumatic central cord syndrome, a retrospective cohort analysis of 50 patients treated acutely (within 24 h) was noted to have shorter intensive care stay, overall length of stay and greater motor improvement (P = 0.04) compared with those decompressed later. This was only noted to be the case in acute fractures or disc herniations. There were no statistitically significant improvements in patients who underwent surgery for cervical stenosis or spondylosis[33].
Another retrospective cohort study of traumatic central cord syndrome, consisting of 126 patients, did not demonstrate any statistically significance difference in neurological recovery based on the timing of surgery. Patients in this cohort treated with surgery did have a shortened length of hospital stay[34].
A prospective cohort analysis of 98 patients with traumatic cervical SCI has showed that early surgical decompression (within 24 h) demonstrated higher rates of ASIA grade recovery post-operatively. At 6 months post-operatively, 23% of the early surgical group had an ASIA grade improvement of at least two grades, compared with 8.7% of the later surgical group[35].
Acute traumatic conus medullaris injury in spinal trauma between levels T12 and L1 has not been shown to have any correlation between neurologic recovery and timing of surgery[7]. The same author performed another retrospective cohort analysis of patients with complete traumatic thoracic SCIs. A cohort of 12 patients showed that in complete thoracic SCIs, two patients demonstrated some sensori-motor improvement, and one patient had motor functional improvement. The median time to surgery in these patients was 11 days, ranging from one to 36 days. In all patients with a documented mechanism of injury, they were all high-energy road traffic accidents, either in vehicles or on motorcycles[6].
However, surgical treatment in traumatic SCI, whether complete or incomplete, is controversial. Conservative management of traumatic SCI has been described in the literature and was utilised to a greater extent in the past. A retrospective cohort analysis published in 1987 reviewed the outcomes of 207 patients with traumatic SCI; 56% of this cohort underwent spinal surgery. There was however, no statistical difference between the patients who underwent surgical treatment versus those managed conservatively in terms of length of stay or neurological recovery[36]. More recently, El Masri et al.[37] discussed the evidence for surgical management compared with Active Physiological Conservative Management. He concluded that conservative, non-surgical management in patients with incomplete SCIs will often recovery enough power to ambulate and suggested the need for review of the current standards of care in relation to the acute management of traumatic SCIs.
The findings of these studies are certainly at odds with the approach to acute traumatic SCI in a study published in 2010. Fehlings et al.[38] assessed the professional opinions and clinical approaches to traumatic SCIs in 972 spinal surgeons. 80% of the surveyed participants would prefer to decompress within 24 h.
Early and aggressive medical management of these patients, followed by appropriate surgical decompression has demonstrated, in prospective studies, improvement of at least one ASIA grade in complete SCIs at one year follow up in 60% of patients. 92% of patients with incomplete cervical spine injuries demonstrated improvement at one-year follow-up in the same study.
Conclusion
SCI carries significant morbidity for affected patients and has a serious economic burden on society. As illustrated by evidence from both the pathophysiology of SCI and clinical outcomes, timing is the key variable that determines treatment outcomes. The acute inflammatory response responsible for demyelination and neuronal damage occurs within minutes of the injury, but peaks at four days after. The vascular and cellular sequelae of these acute inflammatory events, a direct response to trauma and injury, exacerbate the damage and the degree of injury. If clinicians can intervene with the appropriate support to delay, offset or reverse this catastrophic cascade of pro-inflammatory cytokines and ischaemia, patient outcomes will obviously be greatly improved. These interventions must be timely, given how rapidly these celullar responses to injury occur.
Appropriate supportive therapy to maintain mean arterial pressure as described is essential. These patients are often the victims of serious trauma events. As illustrated in the introduction, 40% of these patients are involved in serious road traffic accidents, and may not be suitable for surgical intervention due to concomitant injuries. Coagulopathy, acidosis or renal failure may all render a prolonged surgical intervention dangerous[39-40]. It is in these circumstances that timely and appropriate management from intensivists is paramount. This should, of course, be carried out in a multi-disciplinary manner, with input from local neurosurgical or orthopaedic services.
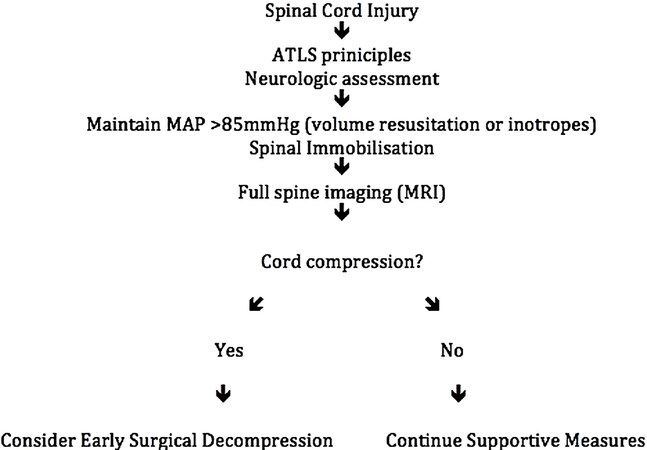
Wherever possible, the prevailing standard of practice appears to guide surgeons to decompress the injured spinal cord within 24 h of injury, particularly in incomplete injuries. This is illustrated by adapated treatment algorithm in Figure 2. This has been shown to offer favourable outcomes in terms of neurologic recovery, compared with delaying surgical intervention. Results are less favourable for patients with complete cord injury. While some studies have offered equivocal results for early decompression, even in incomplete injuries, such as central cord syndromes, the prevailing clinician preference is to decompress as soon as possible.
Figure 2. Treatment algorithm for acute traumatic spinal cord Injury. Adapted from Vale et al.[35]
While there has been a vogue for administering steroids acutely in these patients prior to decompression, they appear to add little in terms of long-term recovery and should be discouraged.
Limitations
Conclusions drawn from this narrative review are derived from a combination of retrospective and prospective cohort analyses, as well as questionnaire publications documenting the treatment preferences of spine surgeons in their treatment protocols. While some evidence cited in this text does reach Level 1, further investigations including randomised controlled trials and meta-analyses would benefit the evidence base in terms of blood pressure management, steroids and surgical decompression.
Summary of recommendations
Patients with acute SCIs should have their mean arterial pressure maintained above 85-90 mmHg after injury for a period of several days;
Where possible, early surgical decompression within 24 h should be undertaken, particularly in incomplete spinal cord injury;
While the timing of establishing musculoskeletal stability of traumatic spinal injuries with surgical decompression and fixation may not determine neurologic return, it certainly allows for earlier mobilisation and easier nursing. This certainly explains the shorter lengths of stay and intensive care stays noted in patients who undergo earlier surgery;
Steroids appear to have no therapeutic value.
Declarations
Authors’ contributionsAll authors contributed equally to the concepts, research, writing and overall academic workload in writing this paper.
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2020.
REFERENCES
1. Mortazavi MM, Verma K, Harmon OA, Griessenauer CJ, Adeeb N, et al. The microanatomy of spinal cord injury: a review. Clin Anat 2015;28:27-36.
2. Gensel JC, Zhang B. Macrophage activation and its role in repair and pathology after spinal cord injury. Brain Res 2015;1619:1-11.
3. Lodish H, Berk A, Zipursky SL, Matsudaira P, Baltimore D, et al. Photosynthetic stages and light-absorbing pigments. Molecular Cell Biology 4th Edition. New York: W. H. Freeman; 2000.
6. Rahimi-Movaghar V, Vaccaro AR, Mohammadi M. Efficacy of surgical decompression in regard to motor recovery in the setting of conus medullaris injury. J Spinal Cord Med 2006;29:32-8.
7. Fleming JC, Norenberg MD, Ramsay DA, Dekaban GA, Marcillo AE, et al. The cellular inflammatory response in human spinal cords after injury. Brain 2006;129:3249-69.
8. Stein DM, Sheth KN. Management of acute spinal cord injury. Continuum (Minneap Minn) 2015;21:159-87.
9. Mautes AE, Weinzierl MR, Donovan F, Noble LJ. Vascular events after spinal cord injury: contribution to secondary pathogenesis. Phys Ther 2000;80:673-87.
10. Ren H, Chen X, Tian M, Zhou J, Ouyang H, et al. Regulation of inflammatory cytokines for spinal cord injury repair through local delivery of therapeutic agents. Adv Sci (Weinh) 2018;5:1800529.
11. Schett G. Physiological effects of modulating the interleukin-6 axis. Rheumatology 2018;57:ii43-50.
12. Yang L, Blumbergs PC, Jones NR, Manavis J, Sarvestani GT, et al. Early expression and cellular localization of proinflammatory cytokines interleukin-1beta, interleukin-6, and tumor necrosis factor-alpha in human traumatic spinal cord injury. Spine (Phila Pa 1976) 2004;29:966-71.
13. Carlson SL, Parrish ME, Springer JE, Doty K, Dossett L. Acute inflammatory response in spinal cord following impact injury. Exp Neurol 1998;151:77-88.
14. Bellver-Landete V, Bretheau F, Mailhot B, Vallières N, Lessard M. Microglia are an essential component of the neuroprotective scar that forms after spinal cord injury. Nat Commun 2019;10:518.
15. Wang H, Liu X, Li R, Zhang P, Chu Z, et al. Effect of glial cells on remyelination after spinal cord injury. Neural Regen Res 2017;12:1724-32.
16. Orr MB, Gensel JC. Spinal cord injury scarring and inflammation: therapies targeting glial and inflammatory responses. Neurotherapeutics 2018;15:541-53.
17. Okada S, Hara M, Kobayakawa K, Matsumoto Y, Nakashima Y. Astrocyte reactivity and astrogliosis after spinal cord injury. Neurosci Res 2018;126:39-43.
18. Gensel JC, Zhang B. Macrophage activation and its role in repair and pathology after spinal cord injury. Brain Res 2015;1619:1-11.
19. Dohrmann GJ, Wagner FC Jr, Bucy PC. Transitory traumatic paraplegia: electron microscopy of early alterations in myelinated nerve fibers. J Neurosurg 1972;36:407-15.
20. Jia Z, Zhu H, Li J, Wang X, Misra H, et al. Oxidative stress in spinal cord injury and antioxidant-based intervention. International Spinal Cord Society. Spinal Cord 2012;50:264-74.
21. Bedreag OH, Rogobete AF, Sărăndan M, Cradigati A, Păpurică M, et al. Oxidative stress and antioxidant therapy in traumatic spinal cord injuries. Rom J Anaesth Intensive Care 2014;21:123-9.
23. Ploumis A, Yadlapalli N, Fehlings MG, Kwon BK, Vaccaro AR. A systematic review of evidence supporting the use of vasopressor support in acute SCI. Spinal Cord 2010;48:356-62.
24. Squair JW, Belanger LM, Tsang A, Ritchie L, Mac-Thiong JM, et al. Spinal cord perfusion pressure predicts neurologic recovery in acute spinal cord injury. Neurology 2017;89:1660-7.
25. Hawryluk G, Whetstone W, Saigal R, Ferguson A, Talbott J, et al. Mean arterial blood pressure correlates with neurological recovery after human spinal cord injury: analysis of high frequency physiologic data. J Neurotrauma 2015;32:1958-67.
26. Dakson A, Brandman D, Thibault-Halman G, Christie SD. Optimization of the mean arterial pressure and timing of surgical decompression in traumatic spinal cord injury: a retrospective study. Spinal Cord 2017;55:1033-8.
27. Saadeh YS, Smith BW, Joseph JR, Jaffer SY, Buckingham MJ, et al. The impact of blood pressure management after spinal cord injury: a systematic review of the literature. Neurosurg Focus 2017;43:E20.
28. Readdy WJ, Whetstone WD, Fergusion AR. Complications and outcomes of vasopressor usage in acute traumatic central cord syndrome. J Neurosurg Spine 2015;23:574-80.
29. Evaniew N, Dvorak M. Cochrane in CORR1: steroids for acute spinal cord injury (Review). Clin Orthop Relat Res 2016;474:19-24.
30. Teles AR, Cabrera J, Riew KD, Falavigna A. Steroid use for acute spinal cord injury in Latin America: a potentially dangerous practice guided by fear of lawsuit. World Neurosurg 2016;88:342-9.
31. Liu JM, Long XU, Zhou Y, Peng HW, Liu ZL, et al. Is urgent decompression superior to delayed surgery for traumatic spinal cord injury? A Meta-Analysis. World Neurosurg 2016;87:124-31.
32. Ter-Wengel PV, De Witt Hamer PC, Pauptit JC, van der Gaag NA, Oner FC, et al. Early surgical decompression improves neurological outcome after complete traumatic cervical spinal cord injury: a meta-analysis. J Neurotrauma 2019;36:835-44.
33. Guest J, Eleraky MA, Apostolides PJ, Dickman CA, Sonntag VK. Traumatic central cord syndrome: results of surgical management. J Neurosurg 2002;97:25-32.
34. Stevens EA, Marsh R, Wilson JA, Sweasey TA, Branch CL Jr, et al. A review of surgical intervention in the setting of traumatic central cord syndrome. Spine J 2010;10:874-80.
35. Vale FL, Burns J, Jackson AB, Hadley MN, et al. Combined medical and surgical treatment after acute spinal cord injury: results of a prospective pilot study to assess the merits of aggressive medical resuscitation and blood pressure management. J Neurosurg 1997;87:239-46.
36. Tator CH, Dunca EG, Edmonds VE, Lapczak LI, Andrews DF. Comparison of surgical and conservative management in 208 patients with acute spinal cord injury. Can J Neurol Sci 1987;14:60-9.
37. El Masri W. Management of traumatic spinal cord injuries: current standard of care revisited. Ad Clin Neuroscience Rehab 2010;10:37-40.
38. Fehlings MG, Rabin D, Sears W, Cadotte DW, Aarabi B. Current practice in the timing of surgical intervention in spinal cord injury. Spine (Phila Pa 1976) 2010;35:S166-73.
39. Mikhail J. The trauma triad of death: hypothermia, acidosis, and coagulopathy. AACN Clin Issues 1999;10:85-94.
40. Germanos S, Gourgiotis S, Villias C. Damage control surgery in the abdomen: an approach for the management of severe injured patients. Int J Surg 2008;6:246-52.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Crozier-Shaw G, Denton H, Morris S. Management strategies in acute traumatic spinal cord injury: a narrative review. Neurosciences 2020;7:335-44. http://dx.doi.org/10.20517/2347-8659.2019.005
AMA Style
Crozier-Shaw G, Denton H, Morris S. Management strategies in acute traumatic spinal cord injury: a narrative review. Neuroimmunology and Neuroinflammation. 2020; 7(4): 335-44. http://dx.doi.org/10.20517/2347-8659.2019.005
Chicago/Turabian Style
Crozier-Shaw, Geoff, Hazel Denton, Seamus Morris. 2020. "Management strategies in acute traumatic spinal cord injury: a narrative review" Neuroimmunology and Neuroinflammation. 7, no.4: 335-44. http://dx.doi.org/10.20517/2347-8659.2019.005
ACS Style
Crozier-Shaw, G.; Denton H.; Morris S. Management strategies in acute traumatic spinal cord injury: a narrative review. Neurosciences. 2020, 7, 335-44. http://dx.doi.org/10.20517/2347-8659.2019.005
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 23 clicks
Cite This Article 23 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.