Use of intravenous immunoglobulin to successfully treat COVID-19 associated encephalitis
Abstract
A 58-year-old male patient presented our emergency department with symptoms of fever and cough of one-week duration. CT scan images of the patient’s chest were suggestive of “Coronavirus disease 2019 Reporting and Data System” score of 5, CT severity score of 16 out of 25, and an oxygen saturation of 95% with a positive reverse transcription polymerase chain reaction test for SARS-CoV-2. The patient was administered medications Remdesivir and Methylprednisolone as per the guidelines. He developed altered sensorium during his stay in the hospital and was electively intubated. MRI of the Brain (Plain and Contrast) showed bilateral cerebellar, parieto-occipital hyperintensities on T2/fluid-attenuated inversion recovery mode. Cerebrospinal fluid (CSF) showed five cells (100% lymphocytes), normal protein and glucose. The patient was non-responsive to the standard COVID management guidelines. In suspicion of viral encephalitis/acute disseminated encephalomyelitis, intravenous immunoglobulins (IVIG) were administered for five days. A steady improvement was observed in the patient’s sensorium over a period of seven days following IVIG therapy, and the patient was able to walk with support following infusion. This case report suggests the possibility of an immune-mediated encephalitis with COVID-19 and the beneficial role of IVIG in treating the same.
Keywords
INTRODUCTION
The SARS-CoV-2 novel Coronavirus presents a range of symptoms varying from running nose to upper and lower respiratory tract infections of variable severity[1]. It is believed that neurological complications can result from SARS-CoV-2 infection based on the knowledge of previous coronavirus epidemics of the severe acute respiratory syndrome and the Middle East respiratory syndrome. Literature on previous coronavirus epidemics of the severe acute respiratory syndrome and the Middle East respiratory syndrome suggests a possible role of SARS-CoV-2 in affecting/involving the neurological system. However, from various literature, it is now evident that COVID-19 can involve multiple organ systems with a specific involvement of the central and peripheral nervous system. Neurological presentations associated with COVID-19 include “anosmia”, “ageusia”, “ataxia”, “seizures”, “hemorrhagic necrotizing encephalopathy” and “Guillain- Barré syndrome”[2-4]. We present a patient of COVID-19 with altered sensorium who improved following administration of intravenous immunoglobulin (IVIG). This supports the evidence of neurotropism of COVID-19. Studies have shown potential use of IVIG in decreasing the respiratory morbidity and mortality in patients with severe COVID-19 infections[5,6].
CASE REPORT
A 58-year-old male patient with type 2 diabetes mellitus and hypertension visited our facility with fever, cough and generalized weakness of one-week duration. As per the COVID-19 guidelines, we administered antivirals (Remedisivir 200 mg loading dose on the 1st day, followed by a 100 mg maintenance dose from the 2nd to 10th day), Methylprednisolone (40 mg intravenous injection twice daily but as cytokine storm set in, the patient received as pulse dose of 1 gram single dose per day for 5 days), and other supportive treatments for COVID-19. The patient developed altered sensorium in the form of picking of clothes, irritability, and shouting from day 3. The patient was agitated, and confused, without following verbal commands. Clinical examination revealed no focal neurological deficits and all the vital signs were normal. The patient was in altered sensorium with eye-opening response only to a deep painful stimulus. He was not obeying our commands and was seen to be anxiously moving limbs against gravity. The pupils were bilaterally equal in size, reacting to light with a positive doll’s eye sign. Deep tendon reflexes were absent upon elicitation. A bilateral plantar withdrawal response was observed. There was no neck stiffness, and the kernig sign was negative. Oxygen saturation was about 98% on the 5 L oxygen mask. Blood pressure recorded was 140/80 mmHg.
CT scan of the chest showed “Coronavirus disease 2019 Reporting and Data System” Score 5 with CT severity score of 16/25[7]. Cerebrospinal fluid (CSF) analysis revealed clear fluid with five cells (100% lymphocytes), normal glucose and protein content. Adenosine deaminase, gram stain and acid fast bacillus were all reported as negative. Steroid dose was increased, and the patient was intubated due to systemic sepsis, hypotension along with a decrease in sensorium. Sepsis could be both due to bacteremia (high serum procalcitonin) and severe viremia. The blood cultures were negative due to early administration of empirical antibiotics.
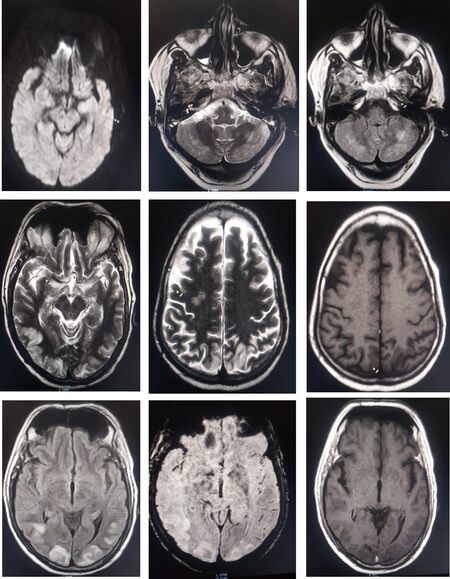
After his hemodynamics were improved, MRI of the Brain (Plain and Contrast) was performed. It showed T2/fluid-attenuated inversion recovery hyperintensities in the bilateral cerebellum, parieto-occipital areas [Figure 1]. Laboratory investigations reported elevated levels of ferritin (828 ng/mL), lactate dehydrogenase (383 U/L), C reactive protein (73.3 mg/L) and D-Dimer (> 1000) [Table 1].
Figure 1. Representative images of the patient showing bilateral cortical/subcortical (temporo-occipital, posterior parietal, right frontoparietal, bilateral cerebellar hemispheres), T2W/FLAIR hyperintensities, with no evidence of diffusion restriction, and no areas of blooming in gradient recalled echo. FLAIR: Fluid-attenuated inversion recovery.
Laboratory values of the patient during the stay in the hospital
| Parameter | Day 1 | Day 2 | Day 6 | Day 8 | Day 10 | Day 12 | Day 15 | At discharge |
| CRP | 93.6 | 50.1 | 12.8 | 22 | 32.3 | 73.3 | 12.8 | 9.5 |
| Serum Ferritin | 348 | 243 | 444 | 489 | 210 | |||
| D Dimer | 355 | 306 | > 1000 | 1038 | 650 | |||
| LDH | 540 | 437 | 675 | 601 | ||||
| IL- 6 | 17.2 | 19 |
Review of recent literature on COVID-19 pertaining to acute disseminated encephalomyelitis and the lack of any clinical improvement with regular treatment, propelled us to discuss IVIG option with the patient attendees and a written informed consent was obtained. Treatment with IVIG (0.4 g/kg) was initiated (32 vials of 10 gram IVIG were given over a period of 5 days). Due to the possible delay in the decrease of viral load, a decision was made to discontinue Methylprednisolone. He progressively improved over the next two days, and the patient was well oriented, able to communicate, and was obeying the commands. The patient recovered entirely by the end of the third day post initiation of IVIG treatment.
The patient was sent home on tracheostomy with a 2 L oxygen mask. Decannulation was done two weeks later. At the end of the first 1-month post treatment, the patient was able to walk without support. MRI Brain repeated was reported as being completely normal except for the periventricular ischemic changes. Two months after discharge, patient returned to near normal activity which was confirmed through telephonic conversation.
DISCUSSION
This report discusses the clinical improvement of encephalitis associated with COVID-19 following administration of IVIG. Considerable variation exists within the available literature on neurologic manifestations of COVID-19. Moriguchi et al.[8] reported first COVID-19 associated meningoencephalitis, which was treated with Favipiravir. Poyiadji et al.[3] reported COVID-19- associated necrotizing encephalopathy with many hemorrhagic rim enhancing lesions. IVIG was the primary treatment modality advocated in the case report with an undefined outcome. Paniz‐Mondolfi et al.[9] reported COVID-19-associated pneumonia with Parkinson’s disease. It was stated in this report that the virus was evident in the capillary endothelium and neuronal cell bodies of the brain. Previously, only one-quarter of the patients with COVID-19 had central nervous system (CNS) manifestations,while Li et al.[2] documented that only about 5% of COVID-19 patients had cerebrovascular events.
As per an earlier report, patients may develop CNS symptoms without associated respiratory symptoms[10]. The interaction with ACE2 receptors,which are abundant in the vascular and neural cells of the CNS, would have allowed the viral particles to enter the CNS[11].
A recent study reported that viral particles were replicating in the endothelial cells of the frontal lobe[9]. This could be attributed to the cytokine storm,which could have altered the blood-brain barrier’s permeability. The possible transmission of corona virus through the olfactory tract could explain the associated anosmia[11]. In our patient, elevated cytokine storm and glial cells’ activation could have resulted in an inflammatory injury that can contribute to encephalitis. CSF polymerase chain reaction testing was not done due to the lack of availability of the facility at our center, because of which SARS-CoV-2 encephalitis could not be ruled out.
The presence of anti-cytokine autoantibodies like anti-TNF-α, anti-IL-1 and anti-IL-8 in natural human immunoglobulin, could be the reason for anti-inflammatory action of IVIG[12]. In addition to this, the natural IgG fraction limits the secretion of proinflammatory cytokines[12]. Dogan et al.[13] reported the potential benefit of plasmapheresis treatment of COVID-19-related autoimmune meningoencephalitis and highlighted the potential role of autoimmunity.
A similar success of IVIG in the treatment of COVID-19 encephalitis was reported previously. However, guidelines on the usage of IVIG in the management of COVID-19 are lacking. IVIG neutralizes activated complement, modulates B and T cells, and inhibits innate immune cells and the release of cytokines[14]. Recovery from encephalitis following administration of IVIG could be attributed to the multiple effects it has on the inflammatory mediators. Many randomized controlled trials on evaluation of efficacy of IVIG in the management of severe COVID-19 currently are in process and the results of these trials are awaited.
DECLARATIONS
Authors’ contributionsConceptualization, review of literature, writing, original draft, reviewing and editing: Chenna V
Writing, original draft, reviewing and editing: Jenjeti C
Investigation, writing, original draft, reviewing and editing: Devarapu K
Availability of data and materialsData are available with corresponding author upon reasonable request.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateInformed consent from participant was obtained.
Consent for publicationConsent for publication was obtained from the participant.
Copyright© The Author(s) 2021.
REFERENCES
1. Zhu N, Zhang D, Wang W, et al. China Novel Coronavirus Investigating and Research Team. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med 2020;382:727-33.
2. Li Y, Li M, Wang M, et al. Acute cerebrovascular disease following COVID-19: a single center, retrospective, observational study. Stroke Vasc Neurol 2020;5:279-84.
3. Poyiadji N, Shahin G, Noujaim D, Stone M, Patel S, Griffith B. COVID-19-associated acute hemorrhagic necrotizing encephalopathy: imaging features. Radiology 2020;296:E119-20.
4. Zhao H, Shen D, Zhou H, Liu J, Chen S. Guillain-Barré syndrome associated with SARS-CoV-2 infection: causality or coincidence? Lancet Neurol 2020;19:383-4.
5. Gharebaghi N, Nejadrahim R, Mousavi SJ, Sadat-Ebrahimi SR, Hajizadeh R. The use of intravenous immunoglobulin gamma for the treatment of severe coronavirus disease 2019: a randomized placebo-controlled double-blind clinical trial. BMC Infect Dis 2020;20:786.
6. Sakoulas G, Geriak M, Kullar R, et al. Intravenous immunoglobulin plus methylprednisolone mitigate respiratory morbidity in coronavirus disease 2019. Crit Care Explor 2020;2:e0280.
7. Wasilewski PG, Mruk B, Mazur S, Półtorak-Szymczak G, Sklinda K, Walecki J. COVID-19 severity scoring systems in radiological imaging - a review. Pol J Radiol 2020;85:e361-8.
8. Moriguchi T, Harii N, Goto J, et al. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int J Infect Dis 2020;94:55-8.
9. Paniz-Mondolfi A, Bryce C, Grimes Z, et al. Central nervous system involvement by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). J Med Virol 2020;92:699-702.
10. Wang HY, Li XL, Yan ZR, Sun XP, Han J, Zhang BW. Potential neurological symptoms of COVID-19. Ther Adv Neurol Disord 2020;13:1756286420917830.
11. Xia H, Lazartigues E. Angiotensin-converting enzyme 2 in the brain: properties and future directions. J Neurochem 2008;107:1482-94.
12. Abe Y, Horiuchi A, Miyake M, Kimura S. Anti-cytokine nature of natural human immunoglobulin: one possible mechanism of the clinical effect of intravenous immunoglobulin therapy. Immunol Rev 1994;139:5-19.
13. Dogan L, Kaya D, Sarikaya T, et al. Plasmapheresis treatment in COVID-19-related autoimmune meningoencephalitis: case series. Brain Behav Immun 2020;87:155-8.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Chenna V, Jenjeti C, Devarapu K. Use of intravenous immunoglobulin to successfully treat COVID-19 associated encephalitis. Neurosciences 2021;8:307. http://dx.doi.org/10.20517/2347-8659.2020.61
AMA Style
Chenna V, Jenjeti C, Devarapu K. Use of intravenous immunoglobulin to successfully treat COVID-19 associated encephalitis. Neuroimmunology and Neuroinflammation. 2021; 8: 307. http://dx.doi.org/10.20517/2347-8659.2020.61
Chicago/Turabian Style
Chenna, Vijay, Chandrasekhar Jenjeti, Katyayani Devarapu. 2021. "Use of intravenous immunoglobulin to successfully treat COVID-19 associated encephalitis" Neuroimmunology and Neuroinflammation. 8: 307. http://dx.doi.org/10.20517/2347-8659.2020.61
ACS Style
Chenna, V.; Jenjeti C.; Devarapu K. Use of intravenous immunoglobulin to successfully treat COVID-19 associated encephalitis. Neurosciences. 2021, 8, 307. http://dx.doi.org/10.20517/2347-8659.2020.61
About This Article
Copyright
Data & Comments
Data
 Cite This Article 15 clicks
Cite This Article 15 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.