MicroRNA-126 is a prospective target for vascular disease
Abstract
MicroRNA-126 was involved in angiogenesis during physiological and pathological process. It was mainly expressed in endothelial cells, and defined as a pivotal biological molecule associated with vascular disease. Increased microRNA-126 in endothelial cells promotes angiogenesis in ischemic stroke, repairs impaired endothelial cells in atherosclerosis, and attenuates vascular dysfunction in diabetics. By contrast, microRNA-126 transferred from endothelial cell to smooth muscle cells could lead to proliferation that induced intimal hyperplasia. Additionally, microRNA-126 could be a tumor suppressor or an oncogene, which was depended on the cancer type. In this review, we summarized the function of microRNA-126 in ischemic stroke, atherosclerosis, diabetics, tumor, and discussed the underlying mechanisms.
Keywords
Introduction
MicroRNAs (miRNAs) are 20-22 nucleotides single-strand and non-coding RNA, which are involved in regulating diverse cellular processes. MiRNAs are originally transcribed from portion of introns of mRNA or independent miRNA genes[1]. MiRNAs regulate gene expression through Ago2-RISC or Ago1-RISC, which depends on if miRNA precisely matched with its target sequence[2].
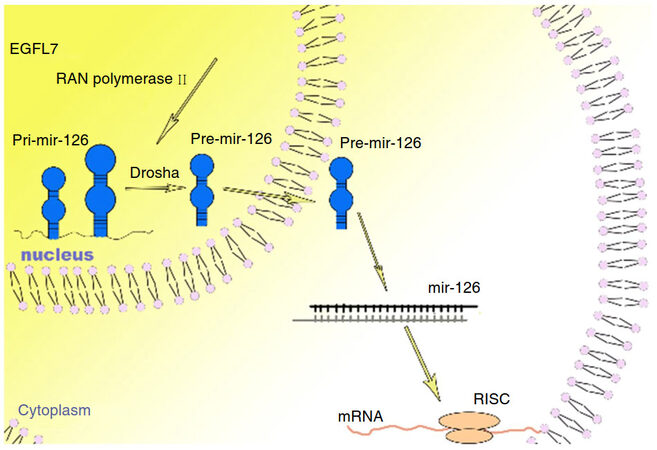
MicroRNA-126 (miR-126) is located in an intron of the epidermal growth factor-like-domain 7 gene (EGFL7). The pri-miR-126 is transcribed from EGFL7 by RNA polymerase II. With the aid of RNase III enzyme Drosha, the pri-miR-126 generates shorter stem-loop precursors (pre-miRNA). Pre-miR-126 is exported to the cytoplasm by exportin-5[3]. In the cytoplasm, RNase III enzyme Dicer processes pre-miR-126 into mature 20-24 nucleotides miRNAs, which then incorporates to the RNA-induced silencing complex (RISC, Figure 1).
Figure 1. MiR-126 is located in the 7th intron of EGFL7. RNA polymerase II binded with EGFL7, resulting in the pri-miR-126 formation. Pri-miRNA-126 targeted to Drosha for cleavage, giving birth to pre-miR-126. Pre-miR-126 was transferred from nucleus to cytoplasm by exporting 5, in which it matured into miR-126. The Dicer cleaved miR-126 into single-strand and either strand of the duplex may potentially act as a functional miRNA, integrating into RISC. The RISC with a microRNA recognized complementary mRNA molecules and degraded or silenced them, resulting in substantially decreased levels of protein translation and effectively inhibiting the gene. EGFL7: epidermal growth factor-like-domain 7 gene; RISC: RNA-induced silencing complex
MiR-126 is specifically and highly expressed in the endothelial cells (ECs), which regulates ECs migration, cytoskeleton reorganization, capillary network stability, cell survival and apoptosis[4]. And microRNA-126 regulates cell survival or apoptosis, depending on different cell types. Furthermore, miR-126 is necessary for the maintenance of vascular structure in vivo[5].
MiR-126 promotes angiogenesis after ischemic stroke
Ischemic stroke was one of the major causes of death and disability in the worldwide[6]. It was classified into large-artery, cardioembolic, and small-vessel (lacunar stroke) ischemic stroke[7]. Currently, angiogenesis was regarded as a promising therapy for the repairing and remodeling after ischemic stroke. Angiogenesis is the growth and remodeling process of the primitive vascular network. Angiogenesis is involved in enlargement of pre-existing vessels or formation of capillaries through trans-endothelial cell bridges[8]. Angiogenesis attenuated functional deficits and promoted behavioral recovery may through restoring the blood flow in the ischemic area. In recent years, studies demonstrated that deletion of miR-126 in mice embryo and zebra fish embryo reduced the integrity of vessels and decreased postnatal angiogenesis[5,9].
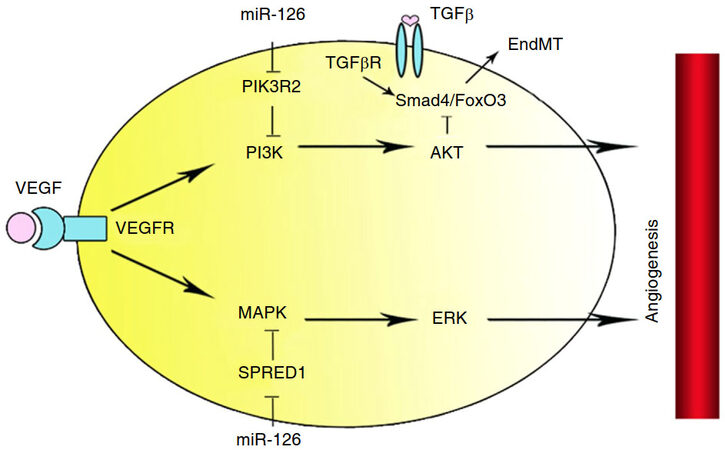
MiR-126 regulated the response of ECs to vascular endothelial growth factor (VEGF), which was via directly repressing negative regulators of VEGF pathway. The negative regulators were the Sprout-related EVH1 domain-containing protein 1 (SPRED1) and phosphoinositol-3 kinase regulatory subunit 2 (PIK3R2)[5]. MiR-126 was an important factor, which could maintain the vascular integrity. Antago-miR-126 decreases ischemia-induced angiogenesis in hind-limb ischemia by increasing SPRED1 and PIK3R2 [Figure 2][10].
Figure 2. In endothelial cells, miR-126 inhibited PIK3R2 and SPRED1 to facilitate angiogenesis by activating PI3K/AKT and MAPK/ERK signal pathway indirectly. In EPCs, miR-126 inhibited EPCs EndMT inducing by TGFb through activating PI3K/AKT signal pathway. EPC: endothelial progenitor cell; VEGF: vascular endothelial growth factor; TGFb: transforming growth factor beta; EndMT: endothelial-to-mesenchymal transition; PIK3R2: phosphoinositol-3 kinase regulatory subunit 2; SPRED1: sprout-related EVH1 domain-containing protein 1
MiR-126 affected the expression of stromal cell derived factor-1 (SDF-1) from different approaches. In normal ECs, miR-126 repressed the SDF-1 synthesis by directly binding to SDF-1 mRNA. Normal miR-126 level was enough to modulate SDF-1 and vascular cell adhesion molecule 1 (VCAM-1) expression in ECs[11]. Under high glucose condition associated with ECs dysfunction, decreasing miR-126 could increase SDF-1 expression, and also directly increased progenitor cells migration and adhesion[11,12], and further improve stroke outcome by differentiating into endothelial cells or through the paracrine effects. Tenreiro et al.[13] demonstrated endothelial cells improved ischemic recovery by differentiation into ECs. Chen et al.[14] demonstrated progenitor cells secreted IL-8 to promote angiogenesis during ischemia. In contrast, under atherosclerosis miR-126 could elevate C-X-C chemokine receptor type 4 (CXCR4) expression by repressing the function of G protein-coupled receptor (GPCR) signaling inhibitor. As a result, SDF-1 could be up-regulated and recruited progenitor cells to the damaged area[15]. In the kidney ischemic condition, miR-126 overexpression in the hematopoietic compartment could attenuate CXCR4 expression on the bone marrow stem cells and at the same time increase SDF-1 in the ischemic tissue. Thus increased SDF-1 facilitated stem cell mobilization towards the ischemic area[16]. Taken together, miR-126 plays a protective role during ischemic injury and is a potential target for ischemic stroke therapy.
MiR-126 plays a dual-role in atherosclerosis
Atherosclerosis was a pathophysiologic process initiated by death of ECs[17]. MiR-126 played a vital role in atherosclerosis[18]. Studies implicated the regulation of ECs proliferation and repair may reduce atherosclerosis formation[19]. In atherosclerosis formation, transferring miR-126 from apoptotic bodies to recipient cells elevated SDF-1, which promoted progenitor cell mobilization and incorporation during plaque formation. Consequently, atherosclerotic progression was impeded[15].
Besides recruiting progenitor cells, miR-126 directly affected ECs proliferation to reduce atherosclerosis[20]. MiR-126-5p promoted ECs proliferation and limits atherosclerosis by suppressing the Noth1 inhibitor delta-like 1 homolog (Dlk1)[20]. MiR-126 down-regulated VCAM-1 expression, thus decreased leukocyte adhesion and resisted vascular wall inflammation[21]. Likewise, up-regulating miR-126 in human aortic ECs (HAEC) showed a decrease of monocyte adhesion via decreasing VCAM-1 expression[22]. In this manner, miR-126 played a beneficial role in atherosclerosis.
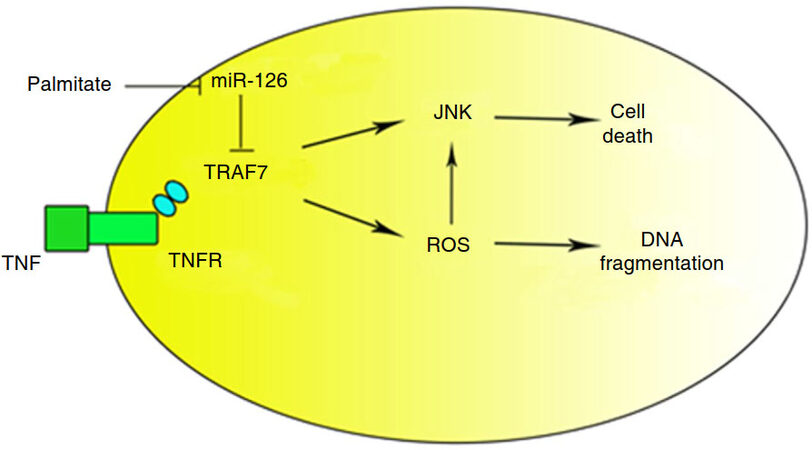
MiR-126 targeted numerous putative mRNAs to exert anti-atherosclerosis effect in varied patterns[18]. For example, it degraded tumor necrosis factor receptor-associated factor 7 (TRAF7) to protect ECs from injury. TRAF7 bound to the tumor necrosis factor receptor (TNFR) and generate intracellular reactive oxygen species (ROS), or reduce the anti-apoptotic molecule expression. MiR-126 was inhibited by palmitate, a major saturated free fatty acid in plasma[23]. MiR-126 overexpression decreased TRAF7 and rescued ECs from saturated free fatty acids (FFAs) damage[23]. These studies demonstrated that miR-126 protected ECs from palmitate and relieved oxidative stress, further induced the effect of anti-atherosclerosis [Figure 3][24].
Figure 3. TRAF7 activated ROS and JNK pathway. MiR-126 increased ROS and inhibited JNK pathway via binding TRAF7. Palmitate promoted cell death and DNA damaged by inhibiting miR-126. TRAF7: tumor necrosis factor receptor-associated factor 7; TNFR: tumor necrosis factor receptor; ROS: reactive oxygen species
In general, endothelial progenitor cells (EPCs) differentiated into ECs to repair damaged intima. However, bone marrow derived EPCs also trans-differentiated into a smooth muscle cell lineage, regarded as endothelial-to-mesenchymal transition (EndMT). Smad3 and Smad4 formed a complex with FoxO3, which played an essential role in cell differentiation. MiR-126 activated PI3K/AKT pathway, and indirectly inhibited FoxO3/Smad4, which was associated with EPC EndMT [Figure 2][25].
Although numerous researches indicated that miR-126 benefited anti-atherosclerosis, studies also demonstrated the adverse effect of miR-126 on atherosclerosis. It was noted that smooth muscle cells (SMCs) could be activated and proliferate in atherosclerosis[26], meanwhile SMC apoptosis caused vulnerable plagues[27]. Transmission of miR-126 to SMCs mediated Forkhead box O3 (FoxO3), B-cell lymphoma-2 (BCL2) and insulin receptor substrate-1 (IRS1) expression, thus affected SMC proliferation, cell cycle progression, and apoptosis[28]. Zhou et al.[28] did not detect microRNA-126 in SMC during atherosclerosis, and a recent study showed that microRNA-126 was up-regulated in atherosclerosis mice[29].
Taken together, miR-126 in SMCs exacerbated atherosclerosis despite it had beneficial effects on endothelial functions. Gene regulation of biological processes extremely complex. Therefore, to explore the modulation mechanism between miRNAs and mRNA seems important.
MiR-126 is a potential target in diabetic ECs dysfunction
Malfunction of ECs was one of the distinct changes in the arterial wall by diabetes induced metabolic abnormalities[30]. Sprout-related EVH1 domain-containing protein 1 (SPRED1) was a Ras/ERK signaling inhibitor, which was involved in the regulation of several cellular processes such as differentiation, survival, motility and cell cycle[31]. MiR-126 affected EPC function via its target SPRED1[31]. PI3K/AKT/eNOS pathway played a role in preventing high glucose-induced cell injury[31]. As mentioned above, miR-126 activated PI3K/AKT/eNOS signal pathway and rescued EPC function by degrading PIK3R[5]. MiR-126 expression was down-regulated in type II diabetic derived EPCs. MiR-126 overexpression in EPCs promoted EPC proliferation, migration, and inhibited EPC apoptosis[31]. Experiments demonstrated that endothelial micro-particles derived from glucose-treated ECs had lower amounts of miR-126, which reduced endothelial repair capacity in vitro and in vivo[32]. It could speculate that miR-126 up-regulation in glucose-damaged ECs protected them from glucose-induced dysfunction.
In consistent with above reports, Zampetaki et al.[33] demonstrated that loss of miR-126 was associated with diabetics. MiR-126 level in endothelial apoptotic bodies was reduced in a glucose-dependent fashion. Low plasma miR-126 level caused VEGF resistance and endothelial dysfunction, which related to diabetics complications[33].
MiR-126 activated VEGF signaling by repressing SPRED1 and PI3R2. Circulating miR-126 has been proposed as a marker for endothelial dysfunction in diabetics. Therefore, up-regulating miR-126 in plasma provided a unique approach for the therapy of endothelial injury.
MiR-126 and tumor
There was no doubt that miR-126 was related to tumorigenic process. Studies showed that MiR-126 was not only a tumor suppressor, but also an oncogene depending on the type of cancer[34-39]. MiR-126 negatively cancer cell proliferation, migration, invasion and survival while it also accelerated cancer progression through the promotion of microvessel formation[34].
Increase of miR-126 was beneficial for the acute myelocytic leukemia (AML) through degrading HOXA9. HOXA9 was an oncogene and often elevated in myelocytic leukemia[35]. However, miR-126 overexpression in AML was associated with poor survival and higher chance of relapse[36]. Decrease of miR-126 expression in AML cells reduced cell growth by inducing apoptosis in vitro[36].
Transferring miR-126 mimics into colon cancer cells reduced cell viability, migration, and invasion by degrading CXCR4[40]. SDF-1 binding to CXCR4 activated NF-kB pathway and increased MMP-2, MMP-9, VEGF and nitric oxide expression. These factors promoted tumor cell invasion through degradation of the extracellular matrix and promotion of angiogenesis, hematopoiesis, ECs growth[41].
MiR-126 is an inhibiting microRNA on the tumor development[38,39]. Studies demonstrated that VEGF-A was a target of MiR-126, which could down-regulate miR-126 and increase VEGF-A expression in tumors[38]. In malignant mesothelioma, miR-126 indirectly increased FOXO1 by targeting IRS1 leading to apoptosis, cell cycle arrest, and stress resistance in various tissues[39].
According to current researches, miR-126 was down-regulated in most tumors such as colorectal cancer, gastric cancer, lung cancer, breast cancer. However, miR-126 was up-regulated in the acute myeloid leukemia[36]. MiR-126 could be a tumor marker in a non-invasive diagnostic method[42].
In conclusion, miR-126 is a double-edged sword and plays distinct roles in different cell types and microenvironment. It could be a potential therapeutic target and prognostic biomarker for the vascular disease, diabetics and tumor. Exploring the effects and underlying mechanisms of miR-126 is important and timely.
Declarations
Authors’ contributionsSummarized the references and wrote the manuscript: Qu MJ
Reviewed literature and discussed paper writing: Pan JJ, Shi XJ
Discussed paper writing and revised the manuscript: Zhang ZJ, Tang YH
Wrote the outline of the paper, edited and approved the final version of the manuscript: Yang GY
Financial support and sponsorshipThis study was supported by grants from the National Key Research and Development Program of China (2016YFC1300600), the National Natural Science Foundation of China (81471178, GYY; 81522015, YTW; 81771251, GYY; 81771244, ZJZ), K. C. Wong Education Foundation (GYY), and the Science and Technology Commission of Shanghai Municipality (17ZR1413600, ZJZ).
Conflicts of interestThere are no conflicts of interest.
Patient consentNot applicable.
Ethics approvalNot applicable.
Copyright© The Author(s) 2018.
REFERENCES
1. Filipowicz W, Bhattacharyya SN, Sonenberg N. Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nat Rev Genet 2008;9:102-14.
2. Forstemann K, Horwich MD, Wee L, Tomari Y, Zamore PD. Drosophila microRNAs are sorted into functionally distinct argonaute complexes after production by dicer-1. Cell 2007;130:287-97.
3. Bohnsack MT, Czaplinski K, Gorlich D. Exportin 5 is a RanGTP-dependent dsRNA-binding protein that mediates nuclear export of pre-miRNAs. RNA 2004;10:185-91.
4. Agrawal S, Chaqour B. MicroRNA signature and function in retinal neovascularization. World J Biol Chem 2014;5:1-11.
5. Fish JE, Santoro MM, Morton SU, Yu S, Yeh RF, Wythe JD, Ivey KN, Bruneau BG, Stainier DYR, Srivastava D. miR-126 regulates angiogenic signaling and vascular integrity. Dev Cell 2008;15:272-84.
6. Kuklina EV, Tong X, George MG, Bansil P. Epidemiology and prevention of stroke: a worldwide perspective. Expert Rev Neurother 2012;12:199-208.
7. Markus HS, Bevan S. Mechanisms and treatment of ischaemic stroke-insights from genetic associations. Nat Rev Neurol 2014;10:723-30.
8. Adams RH, Alitalo K. Molecular regulation of angiogenesis and lymphangiogenesis. Nat Rev Mol Cell Biol 2007;8:464-78.
9. Wang S, Aurora AB, Johnson BA, Qi X, McAnally J, Hill JA, Richardson JA, Bassel-Duby R, Olson EN. The endothelial-specific microRNA miR-126 governs vascular integrity and angiogenesis. Dev Cell 2008;15:261-71.
10. van Solingen C, Seghers L, Bijkerk R, Duijs JM, Roeten MK, van Oeveren-Rietdijk AM, Baelde HJ, Monge M, Vos JB, de Boer HC, Quax PH, Rabelink TJ, van Zonneveld AJ. Antagomir-mediated silencing of endothelial cell specific microRNA-126 impairs ischemia-induced angiogenesis. J Cell Mol Med 2009;13:1577-85.
11. van Solingen C, de Boer HC, Bijkerk R, Monge M, van Oeveren-Rietdijk AM, Seghers L, de Vries MR, van der Veer EP, Quax PH, Rabelink TJ, van Zonneveld AJ. MicroRNA-126 modulates endothelial SDF-1 expression and mobilization of Sca-1(+)/Lin(-) progenitor cells in ischaemia. Cardiovasc Res 2011;92:449-55.
12. Stellos K, Bigalke B, Langer H, Geisler T, Schad A, Kogel A, Pfaff F, Stakos D, Seizer P, Muller I, Htun P, Lindemann S, Gawaz M. Expression of stromal-cell-derived factor-1 on circulating platelets is increased in patients with acute coronary syndrome and correlates with the number of CD34(+) progenitor cells. Eur Heart J 2009;30:584-93.
13. Tenreiro MM, Correia ML, Brito MA. Endothelial progenitor cells in multiple myeloma neovascularization: a brick to the wall. Angiogenesis 2017;20:443-62.
14. Chen C, Lin X, Wang J, Tang G, Mu Z, Chen X, Xu J, Wang Y, Zhang Z, Yang GY. Effect of HMGB1 on the paracrine action of EPC promotes post-ischemic neovascularization in mice. Stem Cells 2014;32:2679-89.
15. Zernecke A, Bidzhekov K, Noels H, Shagdarsuren E, Gan L, Denecke B, Hristov M, Koppel T, Jahantigh MN, Lutgens E, Wang S, Olson EN, Schober A, Weber C. Delivery of microRNA-126 by apoptotic bodies induces CXCL12-dependent vascular protection. Sci Signal 2009;2:ra81.
16. Bijkerk R, van Solingen C, de Boer HC, van der Pol P, Khairoun M, de Bruin RG, van Oeveren-Rietdijk AM, Lievers E, Schlagwein N, van Gijlswijk DJ, Roeten MK, Neshati Z, de Vries AAF, Rodijk M, Pike-Overzet K, van den Berg YW, van der Veer EP, Versteeg HH, Reinders MEJ, Staal FJT, van Kooten C, Rabelink TJ, van Zonneveld AJ. Hematopoietic microRNA-126 protects against renal ischemia/reperfusion injury by promoting vascular integrity. J Am Soc Nephrol 2014;25:1710-22.
17. Xu Q. The impact of progenitor cells in atherosclerosis. Nat Clin Pract Cardiovasc Med 2006;3:94-101.
18. Economou EK, Oikonomou E, Siasos G, Papageorgiou N, Tsalamandris S, Mourouzis K, Papaioanou S, Tousoulis D. The role of microRNAs in coronary artery disease: from pathophysiology to diagnosis and treatment. Atherosclerosis 2015;241:624-33.
19. Davignon J, Ganz P. Role of endothelial dysfunction in atherosclerosis. Circulation 2004;109:III27-32.
20. Schober A, Nazari-Jahantigh M, Wei Y, Bidzhekov K, Gremse F, Grommes J, Megens RT, Heyll K, Noels H, Hristov M, Wang S, Kiessling F, Olson EN, Weber C. MicroRNA-126-5p promotes endothelial proliferation and limits atherosclerosis by suppressing Dlk1. Nat Med 2014;20:368-76.
21. Harris TA, Yamakuchi M, Ferlito M, Mendell JT, Lowenstein CJ. MicroRNA-126 regulates endothelial expression of vascular cell adhesion molecule 1. Proc Natl Acad Sci U S A 2008;105:1516-21.
22. Sun C, Alkhoury K, Wang YI, Foster GA, Radecke CE, Tam K, Edwards CM, Facciotti MT, Armstrong EJ, Knowlton AA, Newman JW, Passerini AG, Simon SI. IRF-1 and miRNA126 modulate VCAM-1 expression in response to a high-fat meal. Circ Res 2012;111:1054-64.
23. Wang Y, Wang F, Wu Y, Zuo L, Zhang S, Zhou Q, Wei W, Wang Y, Zhu H. MicroRNA-126 attenuates palmitate-induced apoptosis by targeting TRAF7 in HUVECs. Mol Cell Biochem 2015;399:123-30.
24. Guo Q, Wang D, He X, Feng Q, Lin R, Xu F, Fu L, Luo M. Whole-brain mapping of inputs to projection neurons and cholinergic interneurons in the dorsal striatum. PLoS One 2015;10:e0123381.
25. Zhang J, Zhang Z, Zhang DY, Zhu J, Zhang T, Wang C. microRNA 126 inhibits the transition of endothelial progenitor cells to mesenchymal cells via the PIK3R2-PI3K/Akt signalling pathway. PLoS One 2013;8:e83294.
26. Imayoshi I, Sakamoto M, Ohtsuka T, Takao K, Miyakawa T, Yamaguchi M, Mori K, Ikeda T, Itohara S, Kageyama R. Roles of continuous neurogenesis in the structural and functional integrity of the adult forebrain. Nat Neurosci 2008;11:1153-61.
27. Clarke MC, Figg N, Maguire JJ, Davenport AP, Goddard M, Littlewood TD, Bennett MR. Apoptosis of vascular smooth muscle cells induces features of plaque vulnerability in atherosclerosis. Nat Med 2006;12:1075-80.
28. Zhou J, Li YS, Nguyen P, Wang KC, Weiss A, Kuo YC, Chiu JJ, Shyy JY, Chien S. Regulation of vascular smooth muscle cell turnover by endothelial cell-secreted microRNA-126: role of shear stress. Circ Res 2013;113:40-51.
29. Hao XZ, Fan HM. Identification of miRNAs as atherosclerosis biomarkers and functional role of miR-126 in atherosclerosis progression through MAPK signalling pathway. Eur Rev Med Pharmacol Sci 2017;21:2725-33.
30. Sena CM, Pereira AM, Seica R. Endothelial dysfunction - a major mediator of diabetic vascular disease. Biochim Biophys Acta 2013;1832:2216-31.
31. Meng S, Cao JT, Zhang B, Zhou Q, Shen CX, Wang CQ. Downregulation of microRNA-126 in endothelial progenitor cells from diabetes patients, impairs their functional properties, via target gene Spred-1. J Mol Cell Cardiol 2012;53:64-72.
32. Jansen F, Yang X, Hoelscher M, Cattelan A, Schmitz T, Proebsting S, Wenzel D, Vosen S, Franklin BS, Fleischmann BK, Nickenig G, Werner N. Endothelial microparticle-mediated transfer of microRNA-126 promotes vascular endothelial cell repair via SPRED1 and is abrogated in glucose-damaged endothelial microparticles. Circulation 2013;128:2026-38.
33. Zampetaki A, Kiechl S, Drozdov I, Willeit P, Mayr U, Prokopi M, Mayr A, Weger S, Oberhollenzer F, Bonora E, Shah A, Willeit J, Mayr M. Plasma microRNA profiling reveals loss of endothelial miR-126 and other microRNAs in type 2 diabetes. Circ Res 2010;107:810-7.
34. Meister J, Schmidt MH. miR-126 and miR-126*: new players in cancer. Scientific World Journal 2010;10:2090-100.
35. Shen WF, Hu YL, Uttarwar L, Passegue E, Largman C. MicroRNA-126 regulates HOXA9 by binding to the homeobox. Mol Cell Biol 2008;28:4609-19.
36. de Leeuw DC, Denkers F, Olthof MC, Rutten AP, Pouwels W, Schuurhuis GJ, Ossenkoppele GJ, Smit L. Attenuation of microRNA-126 expression that drives CD34+38- stem/progenitor cells in acute myeloid leukemia leads to tumor eradication. Cancer Res 2014;74:2094-105.
37. Yan T, Cui K, Huang X, Ding S, Zheng Y, Luo Q, Liu X, Zou L. Assessment of therapeutic efficacy of miR-126 with contrast-enhanced ultrasound in preeclampsia rats. Placenta 2014;35:23-9.
38. Zhu N, Zhang DZ, Xie HP, Zhou Z, Chen HY, Hu TT, Bai YA, Shen YA, Yuan WJ, Jing Q, Qin YW. Endothelial-specific intron-derived miR-126 is down-regulated in human breast cancer and targets both VEGFA and PIK3R2. Mol Cell Biochem 2011;351:157-64.
39. Tomasetti M, Nocchi L, Staffolani S, Manzella N, Amati M, Goodwin J, Kluckova K, Nguyen M, Strafella E, Bajzikova M, Peterka M, Lettlova S, Truksa J, Lee W, Dong LF, Santarelli L, Neuzil J. MicroRNA-126 suppresses mesothelioma malignancy by targeting IRS1 and interfering with the mitochondrial function. Antioxid Redox Signal 2014;21:2109-25.
40. Li Z, Li N, Wu M, Li X, Luo Z, Wang X. Expression of miR-126 suppresses migration and invasion of colon cancer cells by targeting CXCR4. Mol Cell Biochem 2013;381:233-42.
41. Yan T, Liu Y, Cui K, Hu B, Wang F, Zou L. MicroRNA-126 regulates EPCs function: implications for a role of miR-126 in preeclampsia. J Cell Biochem 2013;114:2148-59.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Qu MJ, Pan JJ, Shi XJ, Zhang ZJ, Tang YH, Yang GY. MicroRNA-126 is a prospective target for vascular disease. Neurosciences 2018;5:10. http://dx.doi.org/10.20517/2347-8659.2018.01
AMA Style
Qu MJ, Pan JJ, Shi XJ, Zhang ZJ, Tang YH, Yang GY. MicroRNA-126 is a prospective target for vascular disease. Neuroimmunology and Neuroinflammation. 2018; 5: 10. http://dx.doi.org/10.20517/2347-8659.2018.01
Chicago/Turabian Style
Qu, Mei-Jie, Jia-Ji Pan, Xiao-Jing Shi, Zhi-Jun Zhang, Yao-Hui Tang, Guo-Yuan Yang. 2018. "MicroRNA-126 is a prospective target for vascular disease" Neuroimmunology and Neuroinflammation. 5: 10. http://dx.doi.org/10.20517/2347-8659.2018.01
ACS Style
Qu, M.J.; Pan J.J.; Shi X.J.; Zhang Z.J.; Tang Y.H.; Yang G.Y. MicroRNA-126 is a prospective target for vascular disease. Neurosciences. 2018, 5, 10. http://dx.doi.org/10.20517/2347-8659.2018.01
About This Article
Copyright
Data & Comments
Data
 Cite This Article 22 clicks
Cite This Article 22 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.