Roles of miRNAs in spinal cord injury and potential therapeutic interventions
Abstract
Spinal cord injury (SCI) affects approximately 200,000 individuals per year worldwide. There are more than 27 million people worldwide living with long-term disability due to SCI. Historically, it was thought that the central nervous system (CNS) had little ability for regeneration; however, more recent studies have demonstrated potential for repair within the CNS. Because of this, there exists a renewed interest in the discovery of novel approaches to promote regeneration in the CNS including the spinal cord. It is important to know the roles of the microRNAs (miRNAs) in modulation of pathogenesis in SCI and the potentials of the miRNA-based clinical interventions for controlling post-injury symptoms and improving functional recovery. The miRNAs, which are non-coding RNAs with an average of 22 nucleotides in length, are post-transcriptional gene regulators that cause degradation of the target mRNAs and thus negatively control their translation. This review article focuses on current research related to miRNAs and their roles in modulating SCI symptoms, asserting that miRNAs contribute to critical post-SCI molecular processes including neuroplasticity, functional recovery, astrogliosis, neuropathic pain, inflammation, and apoptosis. In particular, miR-96 provides a promising therapeutic opportunity to improve the outcomes of clinical interventions, including the way SCI injuries are evaluated and treated.
Keywords
Introduction
Spinal cord injury (SCI) results from contusion/compression or transection of the spinal cord. SCI is a significant health issue with an estimate putting the number of people living with this neurological condition at more than 300,000 in the United States[1]. Because of the high number of casualties, SCI is also associated with various socio-economic challenges[2]. Hence, it is essential to understand the molecular mechanisms of pathogenesis in SCI in animal models and to elucidate novel therapeutic interventions for this devastating neurological condition[3]. The emergence of microRNAs (miRNAs) as potent regulators of gene expression at the post-transcriptional level has vast implications in many critical biological processes that include cell proliferation, differentiation, survival, and metabolism[4]. Studies indicate that miRNAs are currently attractive candidates as the upstream regulators of secondary injury progression in SCI because miRNAs are known to regulate entire sets of genes post-transcriptionally. Specific miRNAs (such as miR-96 and miR-544a) are potentially deregulated after SCI and the impact of this deregulation is an area of great interest[5].
Bioinformatic analysis indicates that the potential targets of miRNAs altered after SCI include genes encoding components that are involved in inflammation, oxidative stress, and apoptosis, all of which are known to be crucial for progressive pathogenesis in SCI, suggesting that abnormal expression of miRNAs may contribute to the pathogenesis in SCI. Levels of expression of miRNAs were identified to be deregulated (decreased or increased) in SCI animals[6]. A later investigation showed dramatic decreases in the expression of miRNAs including miR-96 in SCI[7]. Upregulation of miR-96 is likely to promote cell proliferation[8] and prevent neurodegeneration[9] for contribution to functional neuroprotection in SCI. Because miRNAs highly decrease specific gene expression and deregulation of miRNAs does occur in SCI, the potential of particular miRNAs as therapeutic agents should now be explored for functional neuroprotection in SCI[10]. This review article mainly focuses on recent research related to changes in expression of miRNAs following induction of SCI and effects of modulation of levels of miRNAs on critical molecular processes including neuroplasticity, astrogliosis, neuropathic pain, inflammation, apoptosis, and functional recovery in SCI. The last section of this review article focuses explicitly on miR-96 as an emerging post-transcriptional regulator that has the potential to revolutionize SCI clinical interventions.
SCI pathophysiology and miRNAs
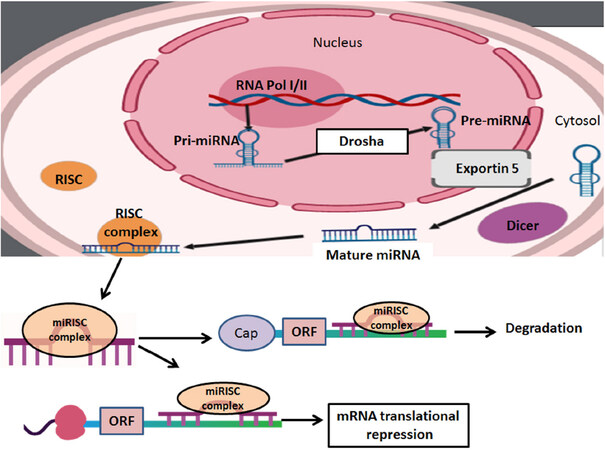
The occurrence of SCI is classified into two different stages: primary stage (a few moments following the initial injury) and secondary stage (hours, days, or weeks after the initial injury). Research shows that the first phase of SCI is the best predictor of future prognosis[11,12]. During the first phase, SCI is manifested with immediate changes in pathophysiology such as hemorrhage, Ca2+ overload, and activation of the Ca2+-dependent cysteine protease calpain causing necrotic and apoptotic neuronal death at the site of impact[13]. The secondary phase begins with molecular and physiological changes responsible for bleeding, loss of neurological functions, expansion of the lesion area, and overall amplification of the injury[14,15]. The secondary phase is also characterized by biochemical reactions, vascular alterations, inflammation, and edema[16]. The main pathological mechanisms responsible for the changes during the secondary phase are the depletion of energy, which is caused by ischemia and oxidative stress, neuroinflammation, and activation of calpains and caspases for cell death at the site of injury and the penumbra[17]. The combination of cellular and molecular modifications leads to various pathological events ranging from astrogliosis to apoptosis and tissue atrophy[18]. Understanding the pathological events is highly crucial for modulating progression of pathogenesis leading to activation of cysteine proteases in both acute and chronic SCI and for implementing therapeutic interventions[19]. It has been shown that attenuation of neuroinflammation and neurodegeneration with appropriate therapeutic interventions is essential for functional recovery in preclinical models of SCI[20,21]. A relatively novel therapeutic intervention evaluated for the treatment of SCI in preclinical models is the utilization of miRNAs[22]. The miRNAs are small non-coding RNA molecules that post-transcriptionally regulate gene expression by degrading and inhibiting translation of target mRNAs [Figure 1].
Figure 1. The biosynthesis of miRNAs begins in the nucleus. RNA polymerase (RNA pol) transcribes primary miRNAs, which consist of a poly-A tail and a 5’ cap. The multi-processor complex made up of double-stranded-RNA-binding protein and the RNase III enzyme Drosha to form pre-miRNAs. The Ran-GTP complex and karyopherin export pre-miRNAs into the cytoplasm. To finalize the process, the RNase III enzyme Dicer cleaves pre-miRNAs and triggers a further processing step by generating the miRNA. When miRNA-induced silencing complex (miRISC) is in the cytoplasm, miRNAs act on the target transcripts via complementary Watson-Crick base pairing to the corresponding miRNA response elements (MREs), which are usually present within the 3ʹ-untranslated regions (3’UTRs) of target genes. Upon binding to MREs within 3’UTRs, miRNAs reduce protein outcome from the target transcripts due to translational repression and/or mRNA deadenylation and decay mechanisms
The biogenesis of miRNAs occurs from pre-miRNA precursors and miRNAs are present not only in body fluid (sputum, serum, and blood) but also in body tissues where they are associated with the regulation of expression of target mRNAs[23]. Different miRNAs are also found in the central nervous system (CNS) where they act as essential mediators of neurodevelopment in the brain and spinal cord in mammals. For example, specific miRNA expression (e.g., miR-17~92 clusters) controls neural growth as well as neurogenesis-gliogenesis switch in the developing CNS including spinal cord. Likewise, miRNAs target genes that are involved in the regulation of essential pathophysiological processes including apoptosis (miR-124 and miR-21), inflammation (miR-544a), and astrogliosis (miR-145 and miR-21) in the spinal cord[24]. The alterations in the expression of miRNAs following SCI can be categorized into three groups: (1) increased miRNAs; (2) decreased miRNAs; and (3) bidirectional (increased or decreased) miRNAs. The expression of specific miRNAs such as miR-146a and miR-129-2 is significantly influenced by the injury severity. The down regulation of specific miRNAs such as miR-219 and miR-124 is associated with the death of neural cells. On the other hand, the overexpression of specific miRNAs (e.g., miR-223) is due to infiltration of vascular and immune cells. Such infiltration of immune cells has been observed during the acute stage of SCI. Research has shown changes in miR-451 during the initial phase of SCI[25]. Other studies also indicate the important roles of miRNAs in regulating immune response and alleviation of inflammation following SCI. For example, miR-544a is down regulated after SCI while genes associated with inflammation (especially NEUROD4) are overexpressed[26]. These results suggest that miR-544a is essential in the repair process after SCI, although further research is required to confirm this assertion.
Potential roles of miRNAs in SCI
miRNAs in astrogliosis
Astrogliosis or astrocytosis refers to the response of astrocytes in response to SCI. Astrogliosis occurs in the area close to the SCI and is a part of a complex multicellular response to SCI. Astrogliosis is characterized by functional, molecular, and morphological changes in astrocytes[27]. These changes usually occur within a few hours following the initial injury and evolve with time[28]. The process of astrogliosis is beneficial in the acute stages where it triggers the repair of the spine-blood barrier, cell regeneration, and prevention of inflammation. However, astrogliosis becomes detrimental in the later stages (4 to 6 weeks) following SCI when astrocytes change from hypertrophic to hyperplastic, producing glial scar with expression of chondroitin sulfate proteoglycans[29].
Under specific pathological conditions, molecular signaling mechanisms associated with miRNAs affects the process of astrocyte proliferation and astrogliosis. For example, the addition of anti-miR-125b is linked with reduced glial cell proliferation and overall regulation of cell growth[30,31]. Similarly, overexpression of miR-145 following SCI has been shown to increase astrogliosis in astrocytes close to the injured area[32]. There was focus of another study, which sought to determine the regulating mechanism of miR-21 in regard to glial scars and astrocytic hypertrophy[33]. More specifically, these investigators carried out tests involving overexpression of miR-21 in mouse astrocytes and concluded that overexpression of miR-21 in astrocytes attenuated hypertrophic response to SCI but expression of the miR-21 sponge augmented hypertrophic phenotype, even in chronic phase of SCI. Other researchers have studied the role of miR-21 in the hypertrophy-hyperplasia shift. According to recent research, miR-21 suppresses the expression of the glial fibrillary acidic protein (GFAP) and vimentin (VIM) under the influence of bone morphogenic protein (BMP) receptors[34]. Activation of different BMP receptors results in differential expression of miR-21 in astrocytes and controls astrogliosis[35,36]. BMP receptor type 1a (BMPR1a) and BMPR1b exert opposite effects on reactive astrocytic hypertrophy. BMPR1b plays a role in glial scar progression in the chronic stages following SCI. These receptors exert opposite effects on expression of miR-21 in astrocytes. Activation of BMPT1a causes overexpression of miR-21 with a dramatic reduction in GFAP levels, limiting the detrimental effects of BMPR1b signaling on glial scar formation following SCI.
miRNAs in apoptosis
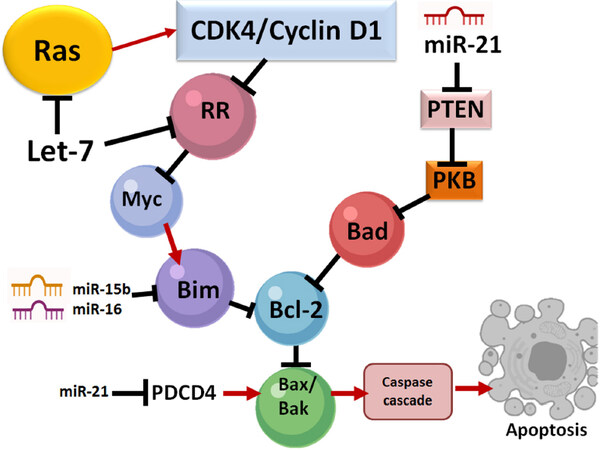
Some researchers studied the action of miRNAs in the context of apoptosis. For example, overexpression of miR-96-5p inhibits apoptosis but promotes migration and proliferation in MDA-MB-231 and MCF-7 cells[37]. In this study, these investigators overexpressed miR-96-5p by transfecting MDA-MB-231 and MCF-7 cells with a miR-96-5p mimic. Treatment and incubation of these cells were done for two days after which cells were double stained with Annexin V/propidium iodide. Flow cytometry was then used to quantify the apoptotic cells. The conceivable molecular events leading to induction of apoptosis in SCI are shown [Figure 2]. Upregulation of miR-96 may suppress programmed cell death protein 4 (PDCD4) and subsequently decrease apoptosis. It is known that reduction of miR-21 promotes apoptosis by inhibiting the expression of phosphatase and tensin homolog (PTEN) and PDCD4[38]. Down regulation of the pro-apoptotic factors PTEN and PDCD4 can increase expression of the cell survival factor Akt or protein kinase B, leading to a reduction in apoptosis in SCI. Studies indicated that specific miRNAs (e.g., miR-21, miR-7-1) could significantly enhance efficacy of promising therapeutic agents (e.g., estrogen receptor agonists) for functional protection of spinal cord motoneurons and this combination therapeutic strategy could be used in the future to attenuate apoptosis of motoneurons in SCI[39,40].
Figure 2. Regulation of apoptosis by miRNAs. All of miR-16, miR-15b, and miR-21 act on several molecular pathways, including Bak, Bcl-2, and Bax pathways during this process. miR-15b, miR-16, and Let-7 down regulate the expression of Bcl-2 (an anti-apoptotic protein) and trigger the release of cytochrome c (a pro-apoptotic factor). Down regulation of Bcl-2 reduces the mitochondrial membrane potential while cytosolic cytochrome c activates the caspase cascade, which subsequently leads to apoptosis. The role of miR-21 is two-fold: protection of neural cells and inhibition of apoptosis. Also, miR-21 down regulates PDCD4 to limit the activation of pro-apoptotic molecular pathways involving Bak and Bax. This, in turn, hinders apoptosis. Also, pro-apoptotic PTEN is down regulated by miR-21, resulting in an anti-apoptotic effect
miRNAs in axon regeneration and remodeling
It has been demonstrated that exercise, which promotes spinal cord plasticity, results in the down regulation of miR-199a-3p and upregulation of miR-21[41]. Alteration in expression of these miRNAs modulates the mechanistic target of rapamycin (mTOR) and PTEN, which are postulated to underlie exercise-induced enhancement of neuronal regeneration in SCI[42]. In another study, it has been shown that upregulation of mTOR occurs by deleting the PTEN gene in mice, resulting in axon regeneration in the injured optic nerves[43,44]. Attenuation of axonal damage and neuronal death is highly crucial for recovery of locomotor function in preclinical models of SCI[45]. Animal model research also suggests that miR-210 carries regenerative properties to cause axon growth in the context of SCI[46]. Administration of miR-210 to SCI mice decreased the expressions of protein-tyrosine phosphate 1B and ephrin-A3 to contribute to spinal cord repair by promoting angiogenesis.
miRNAs in neuronal cell cycle and functional recovery
There is evidence to show that miRNAs not only contribute to regeneration but also stimulate neuron growth and promote functional recovery[47]. Bioinformatic studies propose that miRNAs promote balance between the cell division cycle 42 gene and the brain-derived neurotrophic factor (BDNF) gene, both of which influence self-repair in SCI[48]. Studies show that miR-124 is associated with inhibition of neuronal apoptosis and improvement of motor scores in SCI, with the possibility of even restoring limb functionality after SCI[49]. Study suggests that miR-133b promotes neurite outgrowth via ERK1/2 and PI3K/Akt signaling pathway by RhoA suppression[50]. In terms of functional recovery, evidence shows that miR-133b has the ability to suppress the molecules that inhibit axon regrowth and thereby promote recovery of locomotor function after SCI[51].
miRNAs and neuropathic pain
A rich body of evidence has linked miRNAs to the regulation of SCI-related pain, both neuropathic and inflammatory[52]. According to researchers, the changes in miRNA expression induce increase in insulin-like growth factor-1 expression and down regulation of BDNF. This study concludes that the combination of these changes results in a decrease in inflammation and pain in SCI animals. Down regulation of miR-218 alleviates neuropathic pain by controlling the expression of cytokine signaling, which in turn inhibits the JAK/STAT3 pathway in SCI animals[53]. The expression of miR-124 mainly in neurons is found throughout the brain and spinal cord. A recent study has corroborated the sensitivity of miRNAs to SCI by showing that expression of miR-124 in neurons is significantly decreased within 7 days after SCI to show the severity of injury[54]. The dynamic changes in expression of miRNAs play multiple regulatory mechanisms that may be leveraged to reduce neuropathic pain and potentially shed new light on the progression of maladaptive plasticity in SCI[55].
It has been demonstrated that miR-146a and miR-129-2 regulate pain during the early stages of SCI[56]. In this study, the investigators validated the expression of miRNAs by quantitative reverse transcription-polymerase chain reaction and in situ hybridization assays, revealing that SCI affected miRNA expression that persisted up to 14 days and expanded both anteriorly and caudally beyond the lesion site. It has been suggested that the effect of miRNAs on pain is not necessarily limited to the lesion site. It has been suggested that SCI induces changes in the expression of miRNAs in higher cortical structures, which control neuropathic and inflammatory pain, and miRNAs may serve as specific biomarkers for future targeted therapy of neuropathic and inflammatory pain conditions[57].
Molecular insights into roles of miR-96 in SCI
miR-96 in neuroprotective therapy in SCI
It is essential to mention that miR-96 is one of three miRNAs that make up the miR-183 cluster (the other two miRNAs of this cluster are miR-183 and miR-182)[58]. Recent studies confirm that expression of miR-96 is dramatically decreased after SCI, favoring induction of apoptosis due to increase in expression of its targets, which are pro-apoptotic proteins[59]. Another recent research showed that increase in expression of miR-96 suppressed microglia activation marker proteins and inhibited inflammatory cytokines such as tumor necrosis factor-α and interleukin-1β to promote recovery in SCI[60]. Additionally, it has been reported that miR-96-5p regulates cysteine transporters such as the excitatory amino acid (EAA) transporter 3/EAA carrier 1 (one of the amino acid carriers involved in neuronal glutathione synthesis), which produces neuroprotective benefits against oxidative stress[61]. According to these researchers, rhythmic diurnal fluctuations of glutathione levels occur when miR-96-5p is blocked indirectly influencing the neuroprotective effect[62]. The subsequent section of this article will describe recent research related to roles of miR-96 in inhibition of apoptosis, promotion of cell proliferation, and alteration of other molecular pathways.
miR-96 in inhibition of apoptosis
Research shows that miR-96 may target and inhibit apoptotic factors at the protein and mRNA levels. For instance, miR-96 indirectly inhibits apoptosis through its effect on the forkhead transcription factor of the O class 1 (FOXO1) transcription factor[63]. Studies have shown that FOXO1 induces apoptosis via mitochondria-independent and mitochondria-dependent pathways. Also, miR-96-5p decreases the levels of caspase-9 (an important caspase in the mitochondrial pathway of apoptosis) by binding to the CASP9 3’-untranslated region (3’UTR)[64]. Overexpression of miR-96-5p is associated with inhibition of apoptosis[65].
miR-96 in cell proliferation
There is a growing interest in the study of miR-96, its effect on FOXO1 levels, and how this affects cell proliferation. For example, it has been shown that breast cancer causes upregulation of miR-96 by targeting protein tyrosine phosphatase, non-receptor type 9, this increases cell migration and proliferation and indirectly affects the pathophysiology mechanisms of breast carcinogenesis[66]. Studies on miR-96, specifically its role in hepatocarcinogenesis and treatment of hepatocellular carcinoma (HCC), have shown that miR-96 is significantly upregulated in HCC[67]. Studies also demonstrate that miR-96 targets the FOXO subfamily, specifically FOXO3a and FOXO1. Inhibiting miR-96 upregulates FOXO1 and FOXO3a expression, suppressing colony formation and cell proliferation in HCC[68].
Studies have also shown that prostate cancer is associated with elevated expression of miR-96 and subsequent down regulation of FOXO1, a phenomenon that can be leveraged to control cell proliferation[69]. Other investigators studied the same phenomenon with a dataset containing non-malignant benign prostate tissue samples and prostate cancer tissue samples[70]. According to these researchers, overexpression of miR-96 decreases FOXO1 expression in both the non-malignant tissue samples and the prostate cancer tissue samples, even when the two samples are combined. Down regulation of FOXO3a and p27kip1 promotes axonal regeneration and proliferation of glial cells after SCI in rats[71]. An earlier investigation showed that down regulation of FOXO3a decreased p27kip1 at mRNA and protein levels after injury[72]. A recent study shows that miR-96 is highly essential for normal development of the auditory system, which is required for functional maturation in the peripheral and central auditory system[73].
miR-96 in regulation of FOXO pathway and other molecular pathways
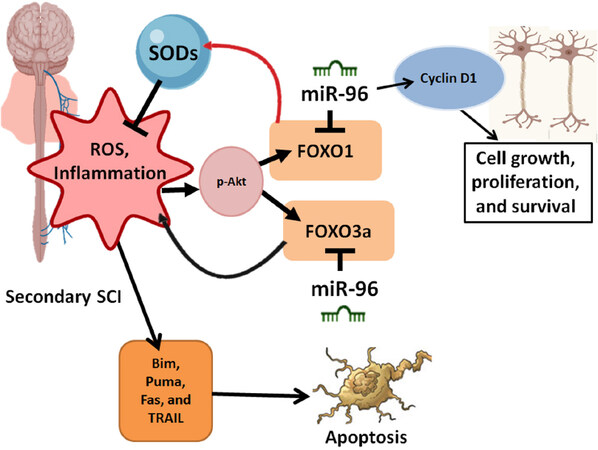
FOXO pathways are of vital importance in regulating biological processes such as glucose metabolism, cellular proliferation, apoptosis, and repair of DNA damage[74]. FOXO transcription factors are also crucial in controlling neurodegenerative disorders through autophagy and apoptosis in the presence of oxidative stress[75]. Mounting evidence demonstrates that miR-96 plays a crucial role in regulating these FOXO pathways. For example, the expression of FOXO3a and FOXO1 is suppressed by miR-96, which controls the cell cycle, cell proliferation, and migration[76]. FOXO3a transcriptionally down regulates the level of p27kip1 (an important neurogenesis regulatory factor in mammals) positioning it as a candidate for controlling axonal regeneration after SCI. The role of miR-96 in regulating FOXO pathways has also been studied in vitro[77], demonstrating that miR-96 binds to the FOXO1 3’UTR sequence lowering the transcript levels of FOXO1 and elevating cell growth [Figure 3].
Figure 3. FOXOs facilitate cell cycle and metabolic regulation, which ultimately regulates apoptosis. At the beginning stage of spinal cord injury (SCI), FOXO1 activates the enzymes (e.g., superoxide dismutases or SODs) responsible for regulating oxidative stress from reactive oxygen species (ROS). With time FOXO1 induces autophagy, controls inflammation, and leads to cell cycle arrest. The role of miR-96 is most profound during the secondary stage of SCI, whereby it regulates FOXO proteins, including FOXO1 to enhances the survival of cells. FOXO3a is also down regulated by miR-96 affecting the expression of inflammatory pathways during SCI. This process also affects pro-apoptotic factors such as TRAIL, Fas, and Puma
Research shows that FOXO3a targets the cyclin-dependent kinase inhibitor p27kip1 and the pro-apoptotic molecule Bim, a phenomenon that triggers apoptosis[78]. Research has also shown that the effect of FOXO3a in regulating apoptosis is dependent on the expression of death receptor ligands such as the FasL[79]. There is evidence showing that miR-96 causes a reduction in the levels of both FOXO1 and FOXO3a for promoting cell proliferation[80], an effect that can be further investigated for neuroprotection and regeneration in SCI.
Conclusion
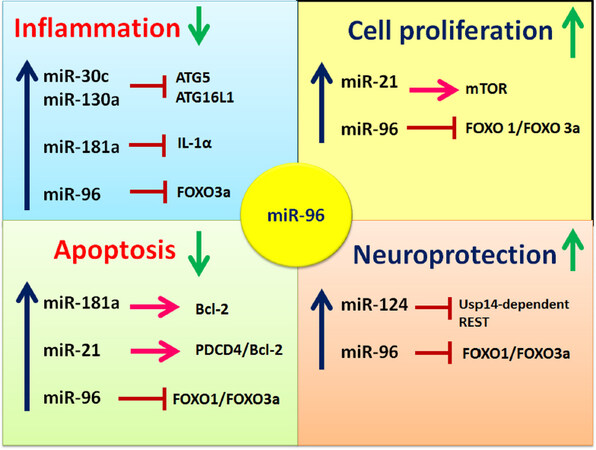
Recent preclinical evidence makes it clear that miRNAs are useful therapeutic tools that modulate critical molecular processes and enhance functional recovery after SCI. In this article, we have discussed some examples, highlighting how the expression of miRNAs triggers complex interactions and changes at the cellular and protein levels. These changes affect SCI pathophysiology and have been identified as regulators and contributors to secondary injury. The assertions made in recent studies indicate that manipulating the expression of miRNAs may provide an opportunity for developing the improved therapeutic and clinical interventions for dealing with the devastating consequences of SCI [Figure 4]. In fact, it is essential to note that not all miRNAs affect SCI positively. However, many of the miRNAs discussed in this review article have been shown to contribute positively to the management of SCI. For example, miR-96 promotes axonal growth, cell regeneration, neuroplasticity, and facilitates functional recovery, although more research is needed in this regard.
Figure 4. Summary of the roles of essential miRNAs including miR-96 in spinal cord injury (SCI). SCI affects the expression of miRNAs, which are known to regulate processes such as cell proliferation, inflammation, and apoptosis. Particularly, miR-96 is essential for neuroprotection as it induces multi-protective functions by targeting and down regulating FOXO pathways
Declarations
Authors’ contributionsConceptualized the theme and conducted the literature review process: Almurshidi B
Contributed to preparation and revision of the manuscript, interpretation of subtopics, and preparation figures: Almurshidi B, Ray SK
Approved the final version to be published: Almurshidi B, Carver W, Scott G, Ray SK
Availability of data and materialsNot applicable.
Financial support and sponsorshipThe work was supported in part by an investigator-initiated research grant (SCIRF-2015-I-01) from South Carolina Spinal Cord Injury Research Fund (Columbia, SC, USA), an award from the Soy Health Research Program (SHRP, United Soybean Board, Chesterfield, MO, USA), and earlier R01 grants (CA-091460 and NS-057811) from the National Institutes of Health (Bethesda, MD, USA).
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2019.
REFERENCES
1. Priebe MM, Chiodo AE, Scelza WM, Kirshblum SC, Wuermser LA, et al. Spinal cord injury medicine. 6. Economic and societal issues in spinal cord injury. Arch Phys Med Rehabil 2007;88:S84-8.
2. Merritt CH, Taylor MA, Yelton CJ, Ray SK. Economic impact of traumatic spinal cord injuries in the United States. Neuroimmunol Neuroinflammation 2019;6:9.
3. Raghava N, Das BC, Ray SK. Neuroprotective effects of estrogen in CNS injuries: insights from animal models. Neurosci Neuroecon 2017;6:15-29.
4. Tafrihi M, Hasheminasab E. miRNAs: biology, biogenesis, their web-based tools, and databases. Microrna 2019;8:4-27.
5. Shi Z, Zhou H, Lu L, Li X, Fu Z, et al. The roles of microRNAs in spinal cord injury. Int J Neurosci 2017;127:1104-15.
6. Liu NK, Wang XF, Lu QB, Xu XM. Altered microRNA expression following traumatic spinal cord injury. Exp Neurol 2009;219:424-9.
7. Yunta M, Nieto-Díaz M, Esteban FJ, Caballero-López M, Navarro-Ruíz R, et al. microRNA dysregulation in the spinal cord following traumatic injury. PLoS One 2012;7:e34534.
8. Ning S, Liu H, Gao B, Wei W, Yang A, et al. miR-155, miR-96 and miR-99a as potential diagnostic and prognostic tools for the clinical management of hepatocellular carcinoma. Oncol Lett 2019;18:3381-7.
9. Loscher CJ, Hokamp K, Wilson JH, Li T, Humphries P, et al. A common microRNA signature in mouse models of retinal degeneration. Exp Eye Res 2008;87:529-34.
10. Li R, Bao L, Hu W, Liang H, Dang X. Expression of miR-210 mediated by adeno-associated virus performed neuroprotective effects on a rat model of acute spinal cord injury. Tissue Cell 2019;57:22-33.
11. James ND, Bartus K, Grist J, Bennett DL, McMahon SB, et al. Conduction failure following spinal cord injury: functional and anatomical changes from acute to chronic stages. J Neurosci 2011;31:18543-55.
12. Silva NA, Sousa N, Reis RL, Salgado AJ. From basics to clinical: a comprehensive review on spinal cord injury. Prog Neurobiol 2014;114:25-57.
13. Ray SK, Hogan EL, Banik NL. Calpain in the pathophysiology of spinal cord injury: neuroprotection with calpain inhibitors. Brain Res Rev 2003;42:169-85.
15. Sezer N, Akkuş S, Uğurlu FG. Chronic complications of spinal cord injury. World J Orthop 2015;6:24-33.
16. Anwar MA, Al Shehabi TS, Eid AH. Inflammogenesis of secondary spinal cord injury. Front Cell Neurosci 2016;10:98.
17. Ray SK, Matzelle DD, Wilford GG, Hogan EL, Banik NL. Inhibition of calpain-mediated apoptosis by E-64-d reduced immediate early gene (IEG) expression and reactive astrogliosis in the lesion and penumbra following spinal cord injury in rats. Brain Res 2001;916:115-26.
18. Faden AI, Wu J, Stoica BA, Loane DJ. Progressive inflammation-mediated neurodegeneration after traumatic brain or spinal cord injury. Br J Pharmacol 2016;173:681-91.
19. Ray SK, Samantaray S, Smith JA, Matzelle DD, Das A, et al. Inhibition of cysteine proteases in acute and chronic spinal cord injury. Neurotherapeutics 2011;8:180-6.
20. Ning B, Gao L, Liu RH, Liu Y, Zhang NS, et al. microRNAs in spinal cord injury: potential roles and therapeutic implications. Int J Biol Sci 2014;10:997-1006.
21. Chakrabarti M, Haque A, Banik NL, Nagarkatti P, Nagarkatti M, et al. Estrogen receptor agonists for attenuation of neuroinflammation and neurodegeneration. Brain Res Bull 2014;109:22-31.
22. Chakrabarti M, Das A, Samantaray S, Smith JA, Banik NL, et al. Molecular mechanisms of estrogen for neuroprotection in spinal cord injury and traumatic brain injury. Rev Neurosci 2016;27:271-81.
23. Slezak-Prochazka I, Durmus S, Kroesen BJ, van den Berg A. microRNAs, macrocontrol: regulation of miRNA processing. RNA 2010;16:1087-95.
24. Madathil SK, Nelson PT, Saatman KE, Wilfred BR. MicroRNAs in CNS injury: potential roles and therapeutic implications. Bioessays 2011;33:21-6.
25. Nieto-Diaz M, Esteban FJ, Reigada D, Muñoz-Galdeano T, Yunta M, et al. MicroRNA dysregulation in spinal cord injury: causes, consequences and therapeutics. Front Cell Neurosci 2014;8:53.
26. Yang L, Ge D, Chen X, Jiang C, Zheng S. miRNA-544a Regulates the Inflammation of Spinal Cord Injury by Inhibiting the Expression of NEUROD4. Cell Physiol Biochem 2018;51:1921-31.
27. Okada S, Hara M, Kobayakawa K, Matsumoto Y, Nakashima Y. Astrocyte reactivity and astrogliosis after spinal cord injury. Neurosci Res 2018;126:39-43.
28. Ito M, Komai K, Mise-Omata S, Iizuka-Koga M, Noguchi Y, et al. Brain regulatory T cells suppress astrogliosis and potentiate neurological recovery. Nature 2019;565:246-50.
29. Karimi-Abdolrezaee S, Billakanti R. Reactive astrogliosis after spinal cord injury-beneficial and detrimental effects. Mol Neurobiol 2012;46:251-64.
30. Pogue AI, Cui JG, Li YY, Zhao Y, Culicchia F, et al. microRNA-125b (miRNA-125b) function in astrogliosis and glial cell proliferation. Neurosci Lett 2010;476:18-22.
31. Pogue AI, Percy ME, Cui JG, Li YY, Bhattacharjee S, et al. Up-regulation of NF-κB-sensitive miRNA-125b and miRNA-146a in metal sulfate-stressed human astroglial (HAG) primary cell cultures. J Inorg Biochem 2011;105:1434-7.
32. Wang CY, Yang SH, Tzeng SF. microRNA-145 as one negative regulator of astrogliosis. Glia 2015;63:194-205.
33. Bhalala OG, Pan L, Sahni V, McGuire TL, Gruner K, et al. microRNA-21 regulates astrocytic response following spinal cord injury. J Neurosci 2012;32:17935-47.
34. Martirosyan NL, Carotenuto A, Patel AA, Kalani MY, Yagmurlu K, et al. The role of microRNA markers in the diagnosis, treatment, and outcome prediction of spinal cord injury. Front Surg 2016;3:56.
35. Sahni V, Mukhopadhyay A, Tysseling V, Hebert A, Birch D, et al. BMPR1a and BMPR1b signaling exert opposing effects on gliosis after spinal cord injury. J Neurosci 2010;30:1839-55.
36. North HA, Pan L, McGuire TL, Brooker S, Kessler JA. β1-Integrin alters ependymal stem cell BMP receptor localization and attenuates astrogliosis after spinal cord injury. J Neurosci 2015;35:3725-33.
37. Shi Y, Zhao Y, Shao N, Ye R, Lin Y, et al. Overexpression of microRNA-96-5p inhibits autophagy and apoptosis and enhances the proliferation, migration and invasiveness of human breast cancer cells. Oncol Lett 2017;13:4402-12.
38. Wang Z, Yao W, Li K, Zheng N, Zheng C, et al. Reduction of miR-21 induces SK-N-SH cell apoptosis and inhibits proliferation via PTEN/PDCD4. Oncol Lett 2017;13:4727-33.
39. Chakrabarti M, Banik NL, Ray SK. miR-7-1 potentiated estrogen receptor agonists for functional neuroprotection in VSC4.1 motoneurons. Neuroscience 2014;256:322-33.
40. Chakrabarti M, Ray SK. Experimental procedures for demonstration of microRNA mediated enhancement of functional neuroprotective effects of estrogen receptor agonists. Methods Mol Biol 2016;1366:359-72.
41. Liu G, Detloff MR, Miller KN, Santi L, Houlé JD. Exercise modulates microRNAs that affect the PTEN/mTOR pathway in rats after spinal cord injury. Exp Neurol 2012;233:447-56.
42. Park KK, Liu K, Hu Y, Kanter JL, He Z. PTEN/mTOR and axon regeneration. Exp Neurol 2010;223:45-50.
43. Sun F, Park KK, Belin S, Wang D, Lu T, et al. Sustained axon regeneration induced by co-deletion of PTEN and SOCS3. Nature 2011;480:372-5.
45. Samantaray S, Sribnick EA, Das A, Knaryan VH, Matzelle DD, et al. Melatonin attenuates calpain upregulation, axonal damage and neuronal death in spinal cord injury in rats. J Pineal Res 2008;44:348-57.
46. Ujigo S, Kamei N, Hadoush H, Fujioka Y, Miyaki S, et al. Administration of microRNA-210 promotes spinal cord regeneration in mice. Spine (Phila Pa 1976) 2014;39:1099-107.
47. Theis T, Yoo M, Park CS, Chen J, Kügler S, et al. Lentiviral delivery of miR-133b improves functional recovery after spinal cord injury in mice. Mol Neurobiol 2017;54:4659-71.
48. Liu Y, Han N, Li Q, Li Z. Bioinformatics analysis of microRNA time-course expression in brown rat (Rattus norvegicus): spinal cord injury self-repair. Spine (Phila Pa 1976) 2016;41:97-103.
49. Yuan S, Wang YX, Gong PH, Meng CY. miR-124 inhibits spinal neuronal apoptosis through binding to GCH1. Eur Rev Med Pharmacol Sci 2019;23:4564-74.
50. Lu XC, Zheng JY, Tang LJ, Huang BS, Li K, et al. miR-133b Promotes neurite outgrowth by targeting RhoA expression. Cell Physiol Biochem 2015;35:246-58.
51. Yu YM, Gibbs KM, Davila J, Campbell N, Sung S, et al. microRNA miR-133b is essential for functional recovery after spinal cord injury in adult zebrafish. Eur J Neurosci 2011;33:1587-97.
52. Strickland ER, Woller SA, Garraway SM, Hook MA, Grau JW, et al. Regulatory effects of intermittent noxious stimulation on spinal cord injury-sensitive microRNAs and their presumptive targets following spinal cord contusion. Front Neural Circuits 2014;8:117.
53. Strickland ER, Woller SA, Hook MA, Grau JW, Miranda RC. The association between spinal cord trauma-sensitive miRNAs and pain sensitivity, and their regulation by morphine. Neurochem Int 2014;77:40-9.
54. Zhao Y, Zhang H, Zhang D, Yu CY, Zhao XH, et al. Loss of microRNA-124 expression in neurons in the peri-lesion area in mice with spinal cord injury. Neural Regen Res 2015;10:1147-52.
55. von Schack D, Agostino MJ, Murray BS, Li Y, Reddy PS, et al. Dynamic changes in the microRNA expression profile reveal multiple regulatory mechanisms in the spinal nerve ligation model of neuropathic pain. PLoS One 2011;6:e17670.
56. Strickland ER, Hook MA, Balaraman S, Huie JR, Grau JW, et al. microRNA dysregulation following spinal cord contusion: implications for neural plasticity and repair. Neuroscience 2011;186:146-60.
57. Andersen HH, Duroux M, Gazerani P. microRNAs as modulators and biomarkers of inflammatory and neuropathic pain conditions. Neurobiol Dis 2014;71:159-68.
58. Banks SA, Pierce ML, Soukup GA. Sensational microRNAs: neurosensory roles of the microRNA-183 family. Mol Neurobiol 2019; doi: 10.1007/s12035-019-01717-3.
59. Pinchi E, Frati A, Cantatore S, D’Errico S, Russa R, et al. Acute spinal cord injury: a systematic review investigating miRNA families involved. Int J Mol Sci ;20:1841.
60. Huang Y, Zhu N, Chen T, Chen W, Kong J, et al. Triptolide suppressed the microglia activation to improve spinal cord injury through miR-96/IKKβ/NF-κB pathway. Spine (Phila Pa 1976) 2019;44:E707-14.
61. Kinoshita C, Aoyama K, Matsumura N, Kikuchi-Utsumi K, Watabe M, et al. Rhythmic oscillations of the microRNA miR-96-5p play a neuroprotective role by indirectly regulating glutathione levels. Nat Commun 2014;5:3823.
62. Kinoshita C, Aoyama K, Nakaki T. Neuroprotection afforded by circadian regulation of intracellular glutathione levels: a key role for miRNAs. Free Radic Biol Med 2018;119:17-33.
63. Guo Y, Liu H, Zhang H, Shang C, Song Y. miR-96 regulates FOXO1-mediated cell apoptosis in bladder cancer. Oncol Lett 2012;4:561-5.
64. Iwai N, Yasui K, Tomie A, Gen Y, Terasaki K, et al. Oncogenic miR-96-5p inhibits apoptosis by targeting the caspase-9 gene in hepatocellular carcinoma. Int J Oncol 2018;53:237-45.
65. Ress AL, Stiegelbauer V, Winter E, Schwarzenbacher D, Kiesslich T, et al. miR-96-5p influences cellular growth and is associated with poor survival in colorectal cancer patients. Mol Carcinog 2015;54:1442-50.
66. Hong Y, Liang H, Uzair-Ur-Rehman, Wang Y, Zhang W, et al. miR-96 promotes cell proliferation, migration and invasion by targeting PTPN9 in breast cancer. Sci Rep 2016;6:37421.
67. Chen RX, Xia YH, Xue TC, Ye SL. Suppression of microRNA-96 expression inhibits the invasion of hepatocellular carcinoma cells. Mol Med Rep 2012;5:800-4.
68. Xu D, He X, Chang Y, Xu C, Jiang X, et al. Inhibition of miR-96 expression reduces cell proliferation and clonogenicity of HepG2 hepatoma cells. Oncol Rep 2013;29:653-61.
69. Haflidadóttir BS, Larne O, Martin M, Persson M, Edsjö A, et al. Upregulation of miR-96 enhances cellular proliferation of prostate cancer cells through FOXO1. PLoS One 2013;8:e72400.
70. Taylor BS, Schultz N, Hieronymus H, Gopalan A, Xiao Y, et al. Integrative genomic profiling of human prostate cancer. Cancer Cell 2010;18:11-22.
71. Zhang S, Huan W, Wei H, Shi J, Fan J, et al. FOXO3a/p27kip1 expression and essential role after acute spinal cord injury in adult rat. J Cell Biochem 2013;114:354-65.
72. Wang Y, Liu Y, Chen Y, Shi S, Qin J, et al. Peripheral nerve injury induces down-regulation of FOXO3a and p27kip1 in rat dorsal root ganglia. Neurochem Res 2009;34:891-8.
73. Schlüter T, Berger C, Rosengauer E, Fieth P, Krohs C, et al. miR-96 is required for normal development of the auditory hindbrain. Hum Mol Genet 2018;27:860-74.
76. Lin H, Dai T, Xiong H, Zhao X, Chen X, et al. Unregulated miR-96 induces cell proliferation in human breast cancer by downregulating transcriptional factor FOXO3a. PLoS One 2010;5:e15797.
77. Song HM, Luo Y, Li DF, Wei CK, Hua KY, et al. microRNA-96 plays an oncogenic role by targeting FOXO1 and regulating AKT/FOXO1/Bim pathway in papillary thyroid carcinoma cells. Int J Clin Exp Pathol 2015;8:9889-900.
78. Yang JY, Xia W, Hu MC. Ionizing radiation activates expression of FOXO3a, Fas ligand, and Bim, and induces cell apoptosis. Int J Oncol 2006;29:643-8.
79. Marfè G, Tafani M, Fiorito F, Pagnini U, Iovane G, et al. Involvement of FOXO transcription factors, TRAIL-FasL/Fas, and sirtuin proteins family in canine coronavirus type II-induced apoptosis. PLoS One 2011;6:e27313.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Almurshidi B, Carver W, Scott G, Ray SK. Roles of miRNAs in spinal cord injury and potential therapeutic interventions. Neurosciences 2019;6:11. http://dx.doi.org/10.20517/2347-8659.2019.19
AMA Style
Almurshidi B, Carver W, Scott G, Ray SK. Roles of miRNAs in spinal cord injury and potential therapeutic interventions. Neuroimmunology and Neuroinflammation. 2019; 6: 11. http://dx.doi.org/10.20517/2347-8659.2019.19
Chicago/Turabian Style
Almurshidi, Badria, Wayne Carver, Geoff Scott, Swapan K. Ray. 2019. "Roles of miRNAs in spinal cord injury and potential therapeutic interventions" Neuroimmunology and Neuroinflammation. 6: 11. http://dx.doi.org/10.20517/2347-8659.2019.19
ACS Style
Almurshidi, B.; Carver W.; Scott G.; Ray SK. Roles of miRNAs in spinal cord injury and potential therapeutic interventions. Neurosciences. 2019, 6, 11. http://dx.doi.org/10.20517/2347-8659.2019.19
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 3 clicks
Cite This Article 3 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.